Advertisements
Advertisements
Question
Draw the geometrical isomers of the following complexes [Pt(NH3)(H2O)Cl2].
Solution
Geometrical isomers of [Pt(NH3)(H2O)Cl2]:
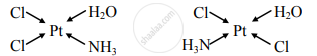
Cis isomer Trans isomer
RELATED QUESTIONS
List various types of isomerism possible for coordination compounds, giving an example of each.
Out of 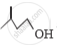 and
and 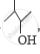 , which one is optically active and why ?
, which one is optically active and why ?
Why dextro and laevo rotatory isomers of Butan-2-ol are difficult to separate by fractional distillation?
Answer the following question.
Draw isomers of the following
Ru(NH3)4Cl2
Which type of isomerism is exhibited by [Pt(NH3)2Cl2]?
What is linkage isomerism? Explain with an example.
Explain optical isomerism in coordination compounds with an example.
What are hydrate isomers? Explain with an example.
Which of the following does NOT show optical isomerism?
____________ isomers are formed when the ligand has two different donor atoms.
Which of the following has zero dipole moment?
How many donor groups are present in diethylene triamine?
What type of isomerism is present between (I) [Cr(H2O)6]Cl3 and (II) [Cr(H2O)5Cl]Cl2.H2O?
\[\ce{IUPAC}\] name of \[\ce{[Pt(NH3)2 Cl(NO2)]}\] is ______.
The correct structure of Fe(CO)5 is
\[\ce{CH3CH2COO- Na+ ->[NaOH, + ?][Heat] CH3CH3 + Na2CO3}\]
Consider the above reaction and identify the missing reagent/chemical.
Write the name of isomerism in the following complexes:
[Cu(NH3)4] [PtCl4] and [Pt(NH3)4] [ CuCl4]
Which compound would exhibit optical isomers?
Which among the following solid is a non-polar solid?
Define Distereoisomers.
Give trans isomer of [Co(NH3)4Cl2]⊕.
Indicate the types of isomerism exhibited by the following complexes and draw the structures for these isomers: [Pt(NH3)(H2O)Cl2]
What are structural isomers or constitutional isomers?
Draw the geometrical isomers of the following complexes [Co(NH3)4Cl2]+
The co-ordination number of Co3+ ion in the complex [Co(NH3)4Cl2]⊕ is ______.
Three organic compounds A, B and C are non cyclic functional isomers of carbonyl compounds with molecular formula C4H8O. Isomers A and C give positive Tollen’s test while compound B does not give positive Tollen’s test but gives positive iodoform test. Compounds A and B on reduction with Zn amalgam and conc. HCl give the same product.
- Write the structures of the compounds A, B and C.
- Out of the compounds A, B and C, which one will be the least reactive towards addition of HCN.
