Advertisements
Advertisements
Question
List various types of isomerism possible for coordination compounds, giving an example of each.
Solution
(i) Geometric isomerism: This type of isomerism is common in heteroleptic complexes. It arises due to the different possible geometric arrangements of the ligands. For example:
\[\begin{array}{cc}
\phantom{..}\ce{Cl}\phantom{.....}\ce{NH3}\phantom{}\\
\backslash\phantom{...}/\\
\ce{Pt}\\
/\phantom{...}\backslash\\
\ce{\underset{cis}{\phantom{..}Cl\phantom{.....}NH3}}
\end{array}\]
\[\begin{array}{cc}
\phantom{..}\ce{Cl}\phantom{.....}\ce{NH3}\phantom{}\\
\phantom{}\backslash\phantom{...}/\\
\ce{Pt}\\
/\phantom{...}\backslash\phantom{}\\
\ce{\underset{trans}{NH3\phantom{....}Cl\phantom{..}}}
\end{array}\]
(ii) Optical isomerism: This type of isomerism arises in chiral molecules. Isomers are mirror images of each other and are non-superimposable.
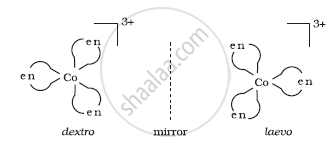
(iii) Linkage isomerism: This type of isomerism is found in complexes that contain ambidentate ligands. For example, [Co(NH3)5(NO2)]Cl2, which is obtained as a red form and [Co(NH3)5(ONO)]Cl2 obtained as a yellow form.
(iv) Coordination isomerism: This type of isomerism arises when the ligands are interchanged between cationic and anionic entities of different metal ions present in the complex. For example, [Co(NH3)6] [Cr(CN)6] and [Cr(NH3)6] [Co(CN)6].
(v) Ionization isomerism: This type of isomerism arises when a counter ion replaces a ligand within the coordination sphere. Thus, complexes that have the same composition but furnish different ions when dissolved in water are called ionization isomers. For example, [Co(NH3)5(SO4)]Br and [Co(NH3)5Br]SO4.
(vi) Solvate isomerism: Solvate isomers differ by whether or not the solvent molecule is directly bonded to the metal ion or merely present as a free solvent molecule in the crystal lattice. For example, [Cr(H2O)6]Cl3 (violet) and [Cr(H2O)5Cl]Cl2.H2O (grey-green).
APPEARS IN
RELATED QUESTIONS
Out of  and
and  , which one is optically active and why ?
, which one is optically active and why ?
Answer the following in one or two sentences.
Predict whether the [Cr(en)2(H2O)2]3+ complex is chiral. Write the structure of its enantiomer.
Answer the following in one or two sentences.
Consider the complexes \[\ce{[Cu(NH3)4][PtCl4] and [Pt(NH3)4] [CuCl4]}\]. What type of isomerism these two complexes exhibit?
Answer in brief.
What are ionization isomers ? Give an example.
The pair [Co(NH3)5(SO4)]Br and [Co(NH3)5Br]SO4 exhibits ____________ isomerism
Which one of the following complexes is not expected to exhibit isomerism?
Fac-mer isomerism is shown by
Draw all possible geometrical isomers of the complex \[\ce{[Co(en)2Cl2]^+}\] and identify the optically active isomer.
____________ isomers are formed when the ligand has two different donor atoms.
The complex ions [Co(H2O)5(ONO)]2+ and [Co(H2O)5NO2]2+ are ____________.
The formula of two complexes X and Y of chromium are given below:
\[\ce{\underset{(X)}{[Cr(H2O)6]Cl3}}\] and \[\ce{\underset{(Y)}{[Cr(H2O)5Cl]Cl2.H2O}}\]
X and Y are examples of ____________ isomers.
Which of the following has zero dipole moment?
Which of the following are isostructural pairs?
(A) \[\ce{SO^{2-}4}\] and \[\ce{CrO^{2-}4}\]
(B) SiCl4 and TiCl4
(C) NH3 and \[\ce{NO^-3}\]
(D) BCl3 and BrCl3
The compounds [PtCl2(NH3)4]Br2 and [PtBr2(NH3)4]Cl2 constitutes a pair of ______.
Explain the ionisation isomers.
The co-ordination number of Co3+ ion in the complex [Co(NH3)4Cl2]⊕ is ______.
Give a chemical test to show that \[\ce{[Co(NH3)5Cl]SO4}\] and \[\ce{[Co(NH3)5SO4]CI}\] are ionisation isomers.
