Advertisements
Advertisements
Question
Answer the following in one or two sentences.
Predict whether the [Cr(en)2(H2O)2]3+ complex is chiral. Write the structure of its enantiomer.
Solution
i. The complex [Cr(en)2(H2O)2]3+ is chiral.
ii. Structures of enantiomers:

APPEARS IN
RELATED QUESTIONS
Write the structural formula and IUPAC names of all possible isomers of the compound with molecular formula C3H8O.
Answer in brief.
What are ionization isomers ? Give an example.
Answer the following question.
Draw isomers of the following
Ru(NH3)4Cl2
Answer the following question.
Draw isomers of the following
[Cr(en2)Br2]⊕
Answer the following question.
Draw geometric isomers and enantiomers of the following complex.
[Pt(en)3]4⊕
Draw optical isomers of [Co(en)3]3+.
Explain optical isomerism in coordination compounds with an example.
Identify the CORRECT statement about the following complex of platinum.
[PtCl2(en)2]2+
Which of the following is NOT an isomer of n-hexane?
Which of the following does NOT show optical isomerism?
____________ isomers are formed when the ligand has two different donor atoms.
The complex ions [Co(H2O)5(ONO)]2+ and [Co(H2O)5NO2]2+ are ____________.
What is the number of moles of silver chloride precipitated when excess of aqueous silver nitrate is treated with [Co(NH3)4Cl2]Cl?
Indicate the complex ion which shows geometrical isomerism.
The relationship between compound (i) and (ii) is
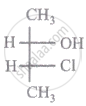 |
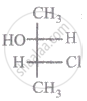 |
| (i) | (ii) |
Which of the following has an optical isomer?
Which compound would exhibit optical isomers?
The number of geometrical isomers of \[\ce{[Co(NH3)3 (NO3)3]}\] are ______.
White precipitate of AgCl dissolves in aqueous ammonia solution due to formation of ______.
The one that is not expected to show isomerism is ______.
Give cis isomer of [Co(NH3)4Cl2]⊕.
Write structures for geometrical isomers of diamminebromochloroplatinum (II).
Draw the structure of trans isomers of Pt(NH3)2Cl2.
The co-ordination number of Co3+ ion in the complex [Co(NH3)4Cl2]⊕ is ______.
Name the type of isomerism exhibited by the following pair of compound:
\[\ce{[Pt(NH3)4Cl2]Br2 and [Pt(NH3)4 Br2]Cl2}\]
Three organic compounds A, B and C are non cyclic functional isomers of carbonyl compounds with molecular formula C4H8O. Isomers A and C give positive Tollen’s test while compound B does not give positive Tollen’s test but gives positive iodoform test. Compounds A and B on reduction with Zn amalgam and conc. HCl give the same product.
- Write the structures of the compounds A, B and C.
- Out of the compounds A, B and C, which one will be the least reactive towards addition of HCN.
