Advertisements
Advertisements
Question
Answer the following question.
Draw isomers of the following
[Cr(en2)Br2]⊕
Solution
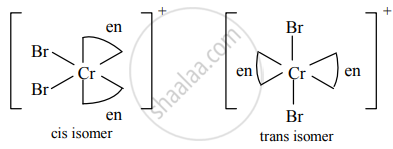
Cis and trans isomers of [Cr(en2)Br2]⊕
a. Cis and trans isomers of [Cr(en2)Br2]+
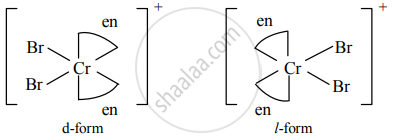
b. Optical isomers of [Cr(en2)Br2]+
APPEARS IN
RELATED QUESTIONS
Choose the most correct option.
Which of the following complexes exist as cis and trans isomers?
1. [Cr(NH3)2Cl4]-
2. [Co(NH3)5Br]2⊕
3. [PtCl2Br2]2- (square planar)
4. [FeCl2(NCS)2]2- (tetrahedral)
Answer in brief.
What are ionization isomers ? Give an example.
Answer the following question.
Draw isomers of the following
Ru(NH3)4Cl2
The pair [Co(NH3)5(SO4)]Br and [Co(NH3)5Br]SO4 exhibits ____________ isomerism
Write the type of isomerism exhibited by [Co(NH3)5(NO2)]2+ and [Co(NH3)5ONO]2+ pair of complex ion.
Draw the geometrical isomers of the following complexes [Pt(NH3)(H2O)Cl2].
Which one of the following pairs represents linkage isomers?
Which one of the following complexes is not expected to exhibit isomerism?
Fac-mer isomerism is shown by
Explain optical isomerism in coordination compounds with an example.
What are hydrate isomers? Explain with an example.
The complex ions [Co(H2O)5(ONO)]2+ and [Co(H2O)5NO2]2+ are ____________.
Indicate the complex ion which shows geometrical isomerism.
\[\ce{IUPAC}\] name of \[\ce{[Pt(NH3)2 Cl(NO2)]}\] is ______.
Geometrical isomerism is not shown by
Which of the following shows maximum number of isomers?
Draw geometric isomers of the following complex.
Geometrical isomers of Pt(NH3)2Cl2
Which of the following compound is optically active?
The one that is not expected to show isomerism is ______.
Which among the following solid is a non-polar solid?
Give cis isomer of [Co(NH3)4Cl2]⊕.
Explain the geometrical isomerism of the octahedral complex of the type [M(AA)2B2]n± with a suitable example.
Draw the structure of trans isomers of Pt(NH3)2Cl2.
The co-ordination number of Co3+ ion in the complex [Co(NH3)4Cl2]⊕ is ______.
Name the type of isomerism exhibited by the following pair of compound:
\[\ce{[Co(NH3)5 [ONO]Cl2 and [Co(NH3)5(NO2)]Cl2}\]
Which one of the following complex ions has geometrical isomers?
