Advertisements
Advertisements
Question
Draw geometric isomers of the following complex.
Geometrical isomers of Pt(NH3)2Cl2
Solution
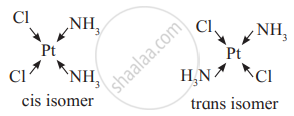
APPEARS IN
RELATED QUESTIONS
List various types of isomerism possible for coordination compounds, giving an example of each.
Why dextro and laevo rotatory isomers of Butan-2-ol are difficult to separate by fractional distillation?
Answer in brief.
What are ionization isomers ? Give an example.
Answer the following question.
Draw isomers of the following
[Cr(en2)Br2]⊕
Answer the following question.
Draw geometric isomers and enantiomers of the following complex.
[Pt(en)3]4⊕
Answer the following question.
Draw geometric isomers and enantiomers of the following complex.
[Pt(en)2ClBr]2⊕
Define the term Hydrated isomers.
Draw the geometrical isomers of the following complexes [Pt(NH3)(H2O)Cl2].
How many geometrical isomers are possible for \[\ce{[Pt(Py)(NH3)(Br)(Cl)]}\]?
Which one of the following pairs represents linkage isomers?
What is linkage isomerism? Explain with an example.
Identify the CORRECT statement about the following complex of platinum.
[PtCl2(en)2]2+
The complex ions [Co(H2O)5(ONO)]2+ and [Co(H2O)5NO2]2+ are ____________.
What is the number of moles of silver chloride precipitated when excess of aqueous silver nitrate is treated with [Co(NH3)4Cl2]Cl?
What type of isomerism is present between (I) [Cr(H2O)6]Cl3 and (II) [Cr(H2O)5Cl]Cl2.H2O?
Indicate the complex ion which shows geometrical isomerism.
Assertion: Complexes of MX6 and MX5L type (X and L are unidentate) do not show geometrical isomerism.
Reason: Geometrical isomerism is not shown by complexes of coordination number 6.
Geometrical isomerism is not shown by
Complex [COCl2(en)2]+ can
Which of the following has an optical isomer?
\[\ce{CH3CH2COO- Na+ ->[NaOH, + ?][Heat] CH3CH3 + Na2CO3}\]
Consider the above reaction and identify the missing reagent/chemical.
Which among the following solid is a non-polar solid?
The compounds [PtCl2(NH3)4]Br2 and [PtBr2(NH3)4]Cl2 constitutes a pair of ______.
Give cis isomer of [Co(NH3)4Cl2]⊕.
Indicate the types of isomerism exhibited by the following complexes and draw the structures for these isomers: [Pt(NH3)(H2O)Cl2]
Draw the geometrical isomers of the following complexes [Co(NH3)4Cl2]+
The co-ordination number of Co3+ ion in the complex [Co(NH3)4Cl2]⊕ is ______.
Name the type of isomerism exhibited by the following pair of compound:
\[\ce{[Co(NH3)5 [ONO]Cl2 and [Co(NH3)5(NO2)]Cl2}\]
