Advertisements
Advertisements
प्रश्न
Answer the following question.
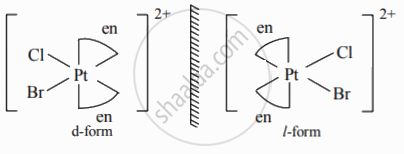
Draw geometric isomers and enantiomers of the following complex.
[Pt(en)2ClBr]2⊕
उत्तर
Geometric isomers of [Pt(en)2ClBr]2⊕:

Enantiomers of [Pt(en)2ClBr]2⊕:
संबंधित प्रश्न
Write the structural formula and IUPAC names of all possible isomers of the compound with molecular formula C3H8O.
Why dextro and laevo rotatory isomers of Butan-2-ol are difficult to separate by fractional distillation?
Choose the most correct option.
Which of the following complexes exist as cis and trans isomers?
1. [Cr(NH3)2Cl4]-
2. [Co(NH3)5Br]2⊕
3. [PtCl2Br2]2- (square planar)
4. [FeCl2(NCS)2]2- (tetrahedral)
Answer in brief.
What are ionization isomers ? Give an example.
Answer the following question.
Draw isomers of the following
[Cr(en2)Br2]⊕
Define the term Co-ordination isomer.
Define the term Hydrated isomers.
Draw the geometrical isomers of the following complexes [Pt(NH3)(H2O)Cl2].
How many geometrical isomers are possible for \[\ce{[Pt(Py)(NH3)(Br)(Cl)]}\]?
Draw all possible geometrical isomers of the complex \[\ce{[Co(en)2Cl2]^+}\] and identify the optically active isomer.
Identify the CORRECT statement about the following complex of platinum.
[PtCl2(en)2]2+
Which of the following is NOT an isomer of n-hexane?
The complex ions [Co(H2O)5(ONO)]2+ and [Co(H2O)5NO2]2+ are ____________.
Indicate the complex ion which shows geometrical isomerism.
Assertion: Complexes of MX6 and MX5L type (X and L are unidentate) do not show geometrical isomerism.
Reason: Geometrical isomerism is not shown by complexes of coordination number 6.
The correct structure of Fe(CO)5 is
Which of the following does not show optical isomerism?
Which of the following has an optical isomer?
Which of the following shows maximum number of isomers?
\[\ce{CH3CH2COO- Na+ ->[NaOH, + ?][Heat] CH3CH3 + Na2CO3}\]
Consider the above reaction and identify the missing reagent/chemical.
White precipitate of AgCl dissolves in aqueous ammonia solution due to formation of ______.
The one that is not expected to show isomerism is ______.
The compounds [PtCl2(NH3)4]Br2 and [PtBr2(NH3)4]Cl2 constitutes a pair of ______.
What are structural isomers or constitutional isomers?
Match the pairs in column I (pairs of isomers) and column II (types of isomers)
| Column I (Pairs of isomers) |
Column II (Types of isomers) |
| (A) [Cr(H2O)5Cl]Cl2.H2O and [Cr(H2O)4Cl2]Cl.2H2O | (i) Ionization isomers |
| (B) [Co(en)2(NO2)2]+ and [Co(en)2(ONO2)]+ | (ii) Hydrate isomers |
| (C) [Co(NH3)6] [Cr(CN)6] and [Cr(NH3)6] [Co(CN)6] | (iii) Linkage isomers |
| (D) [Pt(NH3)4Cl2] Br2 and [Pt(NH3)4Br2]Cl2 | (iv) Coordination isomers |
The co-ordination number of Co3+ ion in the complex [Co(NH3)4Cl2]⊕ is ______.
Name the type of isomerism exhibited by the following pair of compound:
\[\ce{[Co(NH3)5 [ONO]Cl2 and [Co(NH3)5(NO2)]Cl2}\]
