Advertisements
Advertisements
प्रश्न
What is linkage isomerism? Explain with an example.
उत्तर
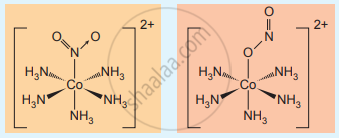
Linkage isomers
Linkage isomerism arises when an ambidentate ligand is bonded to the central metal atom/ion through either of its two different donor atoms.
e.g. - [Co(NH3)5NO2]2+ Pentaammine nitrito-κN Cobalt (III) ion – N attached
[Co(NH3)5ONO2]2+ Pentaammine nitrito-κO Cobalt (III) ion – O attached
APPEARS IN
संबंधित प्रश्न
Write the structural formula and IUPAC names of all possible isomers of the compound with molecular formula C3H8O.
Choose the most correct option.
Which of the following complexes exist as cis and trans isomers?
1. [Cr(NH3)2Cl4]-
2. [Co(NH3)5Br]2⊕
3. [PtCl2Br2]2- (square planar)
4. [FeCl2(NCS)2]2- (tetrahedral)
Answer the following question.
Draw isomers of the following
Ru(NH3)4Cl2
Draw optical isomers of [Co(en)3]3+.
Which one of the following pairs represents linkage isomers?
The compound(s) that exhibit(s) geometrical isomerism is (are):
(I) [Pt(en)Cl2]
(II) [Pt(en)2]Cl2
(III) [Pt(en)2Cl2]
(IV) [Pt(NH3)2Cl2]
Consider the two complexes given below:
\[\ce{\underset{(I)}{[Co(NH3)5SO4]Br}}\] and \[\ce{\underset{(II)}{[Co(NH3)5Br]SO4}}\]
I and II are ____________ isomers.
Write the name of isomerism in the following complexes:
[Cu(NH3)4] [PtCl4] and [Pt(NH3)4] [ CuCl4]
Which compound would exhibit optical isomers?
Draw the geometrical isomers of the following complexes [Co(NH3)4Cl2]+
