Advertisements
Advertisements
प्रश्न
Answer the following question.
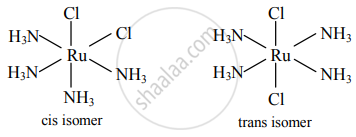
Draw isomers of the following
Ru(NH3)4Cl2
उत्तर
Cis and trans isomers of Ru(NH3)4Cl2
APPEARS IN
संबंधित प्रश्न
List various types of isomerism possible for coordination compounds, giving an example of each.
Give one chemical test as an evidence to show that [Co (NH3)5Cl] are ionisation isomers.
Answer the following in one or two sentences.
Consider the complexes \[\ce{[Cu(NH3)4][PtCl4] and [Pt(NH3)4] [CuCl4]}\]. What type of isomerism these two complexes exhibit?
Answer the following question.
Draw geometric isomers and enantiomers of the following complex.
[Pt(en)3]4⊕
The pair [Co(NH3)5(SO4)]Br and [Co(NH3)5Br]SO4 exhibits ____________ isomerism
Write the type of isomerism exhibited by [Co(NH3)5(NO2)]2+ and [Co(NH3)5ONO]2+ pair of complex ion.
Which type of isomerism is exhibited by [Pt(NH3)2Cl2]?
Which one of the following pairs represents linkage isomers?
Which kind of isomerism is possible for a complex [Co(NH3)4Br2]Cl?
Which of the following is NOT a pair of structural isomers?
How many isomers are possible for an alkane having molecular formula C5H12?
Which of the following does NOT show optical isomerism?
The complex ions [Co(H2O)5(ONO)]2+ and [Co(H2O)5NO2]2+ are ____________.
Which of the following has zero dipole moment?
Which of the following does not show optical isomerism?
Which of the following has an optical isomer?
Write the name of isomerism in the following complexes:
[Cu(NH3)4] [PtCl4] and [Pt(NH3)4] [ CuCl4]
Which compound would exhibit optical isomers?
Which among the following solid is a non-polar solid?
Give trans isomer of [Co(NH3)4Cl2]⊕.
Indicate the type of isomerism exhibited by the following complex and draw the structures for this isomer:
\[\ce{[Pt(NH3)(H2O)Cl2]}\]
Write structures for geometrical isomers of diamminebromochloroplatinum (II).
Explain the geometrical isomerism of the octahedral complex of the type [M(AA)2B2]n± with a suitable example.
Explain the ionisation isomers.
Draw the structure of trans isomers of Pt(NH3)2Cl2.
