Advertisements
Advertisements
प्रश्न
Write the structural formula and IUPAC names of all possible isomers of the compound with molecular formula C3H8O.
उत्तर
| Sr. no. | Structural Formula | IUPAC Name |
| 1. | CH3 - CH2 - CH2 - OH | Propan-1-ol |
| 2. | \[\begin{array}{cc} \ce{CH3 - CH - CH3}\\ |\phantom{..}\\ \ce{OH} \end{array}\] |
Propan-2-ol |
| 3. | CH3CH2 - O - CH3 | 1-Methoxyethane |
APPEARS IN
संबंधित प्रश्न
List various types of isomerism possible for coordination compounds, giving an example of each.
Answer the following in one or two sentences.
Consider the complexes \[\ce{[Cu(NH3)4][PtCl4] and [Pt(NH3)4] [CuCl4]}\]. What type of isomerism these two complexes exhibit?
Answer the following question.
Draw isomers of the following
[Cr(en2)Br2]⊕
The pair [Co(NH3)5(SO4)]Br and [Co(NH3)5Br]SO4 exhibits ____________ isomerism
Define the term Co-ordination isomer.
Which one of the following pairs represents linkage isomers?
Explain optical isomerism in coordination compounds with an example.
The term anomers of glucose refer to ____________.
The compound(s) that exhibit(s) geometrical isomerism is (are):
(I) [Pt(en)Cl2]
(II) [Pt(en)2]Cl2
(III) [Pt(en)2Cl2]
(IV) [Pt(NH3)2Cl2]
The number of geometrical isomers of [CrCl2(en)2]+ is ____________.
The formula of two complexes X and Y of chromium are given below:
\[\ce{\underset{(X)}{[Cr(H2O)6]Cl3}}\] and \[\ce{\underset{(Y)}{[Cr(H2O)5Cl]Cl2.H2O}}\]
X and Y are examples of ____________ isomers.
What type of isomerism is present between (I) [Cr(H2O)6]Cl3 and (II) [Cr(H2O)5Cl]Cl2.H2O?
The relationship between compound (i) and (ii) is
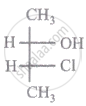 |
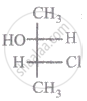 |
| (i) | (ii) |
Geometrical isomerism is not shown by
Complex [COCl2(en)2]+ can
Write the name of isomerism in the following complexes:
[Cu(NH3)4] [PtCl4] and [Pt(NH3)4] [ CuCl4]
Draw geometric isomers of the following complex.
Geometrical isomers of Pt(NH3)2Cl2
Which of the following are isostructural pairs?
(A) \[\ce{SO^{2-}4}\] and \[\ce{CrO^{2-}4}\]
(B) SiCl4 and TiCl4
(C) NH3 and \[\ce{NO^-3}\]
(D) BCl3 and BrCl3
Which compound would exhibit optical isomers?
The one that is not expected to show isomerism is ______.
Which among the following solid is a non-polar solid?
Draw the structure of trans isomers of Pt(NH3)2Cl2.
Name the type of isomerism exhibited by the following pair of compound:
\[\ce{[Co(NH3)5 [ONO]Cl2 and [Co(NH3)5(NO2)]Cl2}\]
Name the type of isomerism exhibited by the following pair of compound:
\[\ce{[Pt(NH3)4Cl2]Br2 and [Pt(NH3)4 Br2]Cl2}\]
Give a chemical test to show that \[\ce{[Co(NH3)5Cl]SO4}\] and \[\ce{[Co(NH3)5SO4]CI}\] are ionisation isomers.
Three organic compounds A, B and C are non cyclic functional isomers of carbonyl compounds with molecular formula C4H8O. Isomers A and C give positive Tollen’s test while compound B does not give positive Tollen’s test but gives positive iodoform test. Compounds A and B on reduction with Zn amalgam and conc. HCl give the same product.
- Write the structures of the compounds A, B and C.
- Out of the compounds A, B and C, which one will be the least reactive towards addition of HCN.
