Advertisements
Advertisements
प्रश्न
Answer the following questions based on the P–T phase diagram of CO2:
CO2 at 1 atm pressure and temperature – 60 °C is compressed isothermally. Does it go through a liquid phase?
उत्तर १
No
The P-T phase diagram for CO2 is shown in the following figure.
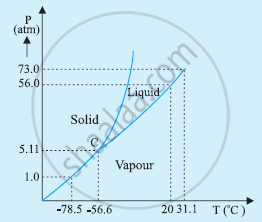
At 1 atm pressure and at –60°C, CO2 lies to the left of –56.6°C (triple point C). Hence, it lies in the region of vaporous and solid phases.
Thus, CO2 condenses into the solid state directly, without going through the liquid state
उत्तर २
No, the CO2 does not go through the liquid phase. The point (1.00 atm, – 60°C) is to the lift of the triple-point O and below the sublimation curve OA. Therefore, when CO2 is compressed at this point at constant temperature, the point moves perpendicular to the temperature-axis and enters the solid phase region. Hence, the CO2 vapour condenses to solid directly without going through the liquid phase.
APPEARS IN
संबंधित प्रश्न
Answer the following questions based on the P–T phase diagram of CO2:
What happens when CO2 at 4 atm pressure is cooled from room temperature at constant pressure?
Answer the following questions based on the P–T phase diagram of CO2:
Describe qualitatively the changes in a given mass of solid CO2 at 10 atm pressure and temperature –65 °C as it is heated up to room temperature at constant pressure.
Answer the following questions based on the P–T phase diagram of CO2:
Describe qualitatively the changes in a given mass of solid CO2 at 10 atm pressure and temperature –65 °C as it is heated up to room temperature at constant pressure.
Answer the following questions based on the P–T phase diagram of CO2:
CO2 is heated to a temperature 70 °C and compressed isothermally. What changes in its properties do you expect to observe?
Explain why a body with large reflectivity is a poor emitter
Answer the following question based on the P-T phase diagram of carbon dioxide:
What is the effect of decrease of pressure on the fusion and boiling point of CO2?
Answer the following question based on the P-T phase diagram of carbon dioxide:
What are the critical temperature and pressure for CO2? What is their significance?
The radii of two copper spheres are in the ratio 1 : 3 and their temperatures are in the ratio 9 : 1. The ratio of heat content in them will be:
Mark the correct options:
- A system X is in thermal equilibrium with Y but not with Z. System Y and Z may be in thermal equilibrium with each other.
- A system X is in thermal equilibrium with Y but not with Z. Systems Y and Z are not in thermal equilibrium with each other.
- A system X is neither in thermal equilibrium with Y nor with Z. The systems Y and Z must be in thermal equilibrium with each other.
- A system X is neither in thermal equilibrium with Y nor with Z. The system Y and Z may be in thermal equilibrium with each other.
During summers in India, one of the common practice to keep cool is to make ice balls of crushed ice, dip it in flavoured sugar syrup and sip it. For this a stick is inserted into crushed ice and is squeezed in the palm to make it into the ball. Equivalently in winter, in those areas where it snows, people make snow balls and throw around. Explain the formation of ball out of crushed ice or snow in the light of P–T diagram of water.
