Advertisements
Advertisements
प्रश्न
Complete and balance the following chemical equations:
`NaHCO_3` 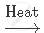
उत्तर
`2NaHCO_3` `Na_2 CO_3 + CO_2 + H_2O`
`Na_2 CO_3 + CO_2 + H_2O`
APPEARS IN
संबंधित प्रश्न
When you add sodium hydrogen carbonate to acetic acid in a test tube, a gas liberates immediately with brisk effervescence. Name this gas. Describe the method of testing this gas.
The discomfort caused by indigestion due to overeating can be cured by taking:
Which compound is used as an antacid in medicine : NaHCO3 or Na2CO3?
What happens when a cold and concentrated solution of sodium chloride reacts with ammonia and carbon dioxide? Write the chemical equation of the reaction which takes place.
Consider the following substances:
NaCl, Ca(OH)2, NaHCO3, NH3, Na2CO3, H2O, Cl2, CO2, CaSO4.2H2O, 2CaSO4.H2O, CaOCl2
Which compound is a part of baking powder?
A person found that the cake prepared by him is hard and small in size. Which ingredient has he forgotten to add that would have caused the cake to rise and become light? Explain your answer.
(i) Name the products formed when sodium hydrogen carbonate is heated.
(ii) Write the chemical equation for the reaction involved in the above.
Baking soda is a mixture of:
For making cake, baking powder is taken. If at home your mother uses baking soda instead of baking powder in cake,
- how will it affect the taste of the cake and why?
- how can baking soda be converted into baking powder?
- what is the role of tartaric acid added to baking soda?
Mild non-corrosive basic salt is ______.
