Advertisements
Advertisements
प्रश्न
When you add sodium hydrogen carbonate to acetic acid in a test tube, a gas liberates immediately with brisk effervescence. Name this gas. Describe the method of testing this gas.
List two observations which you make when you add a pinch of sodium hydrogen carbonate to acetic acid in a test tube. Write chemical equation for the reaction that occurs.
उत्तर
Carbon dioxide gas gets liberated.
When a pinch of sodium hydrogen carbonate is added to acetic acid in a test tube, a brisk effervescence is produced because of the liberation of carbon dioxide gas.
When this gas is passed through the lime water, it turns lime water milky. This shows that the gas liberated is carbon dioxide gas.
The chemical reaction can be represented as
CH3COOH(aq) + NaHCO3(S) → CH3COONa(aq) + H2O(l) + CO2(g) ↑
APPEARS IN
संबंधित प्रश्न
The discomfort caused by indigestion due to overeating can be cured by taking:
Complete and balance the following chemical equations:
`NaHCO_3` 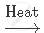
What is baking soda?
What happens when a cold and concentrated solution of sodium chloride reacts with ammonia and carbon dioxide? Write the chemical equation of the reaction which takes place.
What is the common name of sodium hydrogencarbonate?
How does baking powder differ from baking soda?
The chemical name of baking soda is ______.
NaHCO, formed by reaction of ____________.
For making cake, baking powder is taken. If at home your mother uses baking soda instead of baking powder in cake,
- how will it affect the taste of the cake and why?
- how can baking soda be converted into baking powder?
- what is the role of tartaric acid added to baking soda?
Mild non-corrosive basic salt is ______.
