Advertisements
Advertisements
प्रश्न
Complete the following equations:
KMnO4 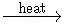
उत्तर
2 KMnO4  K2MnO4 + MnO2 + O2
K2MnO4 + MnO2 + O2
APPEARS IN
संबंधित प्रश्न
Account for the following: Fluorine does not exhibit positive oxidation state.
Compare the oxidizing action of F2 and Cl2 by considering parameters such as bond dissociation enthalpy, electron gain enthalpy and hydration enthalpy.
F2 has lower bond dissociation enthalpy than Cl2. Why?
Write two uses of ClO2.
Write the reactions of F2 and Cl2 with water.
Arrange the following in the order of property indicated for each set:
HF, HCl, HBr, HI - increasing acid strength.
Select the correct alternative from the choices given :
The correct order of increasing acidic strength of the oxoacids of chlorine is:
Give reason to explain why ClF3 exists but FCl3 does not exist.
Iodine has lowest oxidiation state in ______.
Statement I: Acid strength increases in the order given as HF << HCl << HBr << HI.
Statement II: As the size of the elements F, Cl, Br, and I increase down the group, the bond strength of HF, HCl, HBr and HI decreases and so the acid strength increases.
In light of the above statements, choose the correct answer from the options given below.
