Advertisements
Advertisements
Question
Complete the following equations:
KMnO4 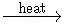
Solution
2 KMnO4  K2MnO4 + MnO2 + O2
K2MnO4 + MnO2 + O2
APPEARS IN
RELATED QUESTIONS
Draw structure of Chlorine pentafluoride
Account for the following :
Acidic character increases from HF to HI.
F2 has lower bond dissociation enthalpy than Cl2. Why?
Which halogen compound in each of the following pairs will react faster in SN2 reaction
(CH3)3 C – Cl or CH3 – Cl
Why are halogens coloured?
Arrange the following in the decreasing order of their reducing character :
HF, HCl, HBr, HI
Cl2 acts as a bleaching agent.
Reduction potentials of some ions are given below. Arrange them in decreasing order of oxidising power.
| Ion | \[\ce{CIO^{-}_{4}}\] | \[\ce{IO^{-}_{4}}\] | \[\ce{BrO^{-}_{4}}\] |
| Reduction potential EΘ/V |
EΘ = 1.19 V | EΘ = 1.65V | EΘ = 1.74 V |
The reaction \[\ce{3ClO- (aq) -> ClO3- (aq) + 2Cl- (aq) + 2Cl- (aq)}\] is an example of
Give a reason for the following:
Mn+2 compounds are more stable than Fe+2 compounds.
