Advertisements
Advertisements
प्रश्न
Give reason to explain why ClF3 exists but FCl3 does not exist.
उत्तर
Chlorine has vacant d-orbitals, which become excited upon bonding when electrons from the 3p-orbital are promoted to the 3d-orbital, giving it a covalency of three.
Due to the lack of unoccupied d-orbitals in the second energy shell, fluorine cannot expand its octet. As a result, it can only have one covalency.
APPEARS IN
संबंधित प्रश्न
Compare the oxidizing action of F2 and Cl2 by considering parameters such as bond dissociation enthalpy, electron gain enthalpy and hydration enthalpy.
Give two examples to show the anomalous behaviour of fluorine.
Why are halogens coloured?
Complete the following equations:
KMnO4 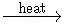
Which one has the lowest boiling point?
In solid state \[\ce{PCl5}\] is a ______.
Reduction potentials of some ions are given below. Arrange them in decreasing order of oxidising power.
| Ion | \[\ce{CIO^{-}_{4}}\] | \[\ce{IO^{-}_{4}}\] | \[\ce{BrO^{-}_{4}}\] |
| Reduction potential EΘ/V |
EΘ = 1.19 V | EΘ = 1.65V | EΘ = 1.74 V |
Solubility of iodine in water may be increase by adding
Give a reason for the following:
Mn+2 compounds are more stable than Fe+2 compounds.
Arrange the following in the increasing order of the property mentioned:
\[\ce{MF}\], \[\ce{MCl}\], \[\ce{MBr}\], \[\ce{MI}\] (ionic character)
