Advertisements
Advertisements
प्रश्न
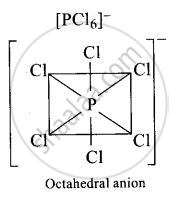
In solid state \[\ce{PCl5}\] is a ______.
पर्याय
covalent solid
octahedral structure
ionic solid with \[\ce{[PCl_{6}]^{+}}\] octahedral and \[\ce{[PCl_{4}]^{-} tetrahedra}\]
ionic solid with \[\ce{[PCl_{4}]^{+}}\] octahedral and \[\ce{[PCl_{6}]^{-} tetrahedra}\]
उत्तर
In solid state \[\ce{PCl5}\] is a ionic solid with \[\ce{[PCl_{4}]^{+}}\] octahedral and \[\ce{[PCl_{6}]^{-} tetrahedra}\].
Explanation:

APPEARS IN
संबंधित प्रश्न
Which halogen compound in each of the following pairs will react faster in SN2 reaction CH3Br or CH3I
Which halogen compound in each of the following pairs will react faster in SN2 reaction
(CH3)3 C – Cl or CH3 – Cl
Give two examples to show the anomalous behaviour of fluorine.
Write the reactions of F2 and Cl2 with water.
Tincture of iodine is ____________.
The set with the correct order of acidity is:
Which is the strongest acid in the following:
Reduction potentials of some ions are given below. Arrange them in decreasing order of oxidising power.
| Ion | \[\ce{CIO^{-}_{4}}\] | \[\ce{IO^{-}_{4}}\] | \[\ce{BrO^{-}_{4}}\] |
| Reduction potential EΘ/V |
EΘ = 1.19 V | EΘ = 1.65V | EΘ = 1.74 V |
Give reason to explain why ClF3 exists but FCl3 does not exist.
Give a reason for the following:
Mn+2 compounds are more stable than Fe+2 compounds.
