Advertisements
Advertisements
प्रश्न
Compounds such as alcohol and glucose also contain hydrogen but are not categorised as acids. Describe an activity to prove it.
उत्तर
Activity to show that alcohol and glucose also contain hydrogen but are not acids:
Procedure:
i) Take samples of alcohol, glucose and hydrochloric acid.
ii) Take a 100 ml beaker. Take a cork and fix two nails on it.
iii) Place the cork inside the beaker as shown in the figure.
iv) Connect the nails to two terminals of a 6-volt battery through a bulb and a switch.
v) Now pour some dilute hydrochloric acid in the beaker so that the nails and the cork are immersed in it.
vi) Switch on the current flow.
vii) Repeat the same with alcohol and glucose.
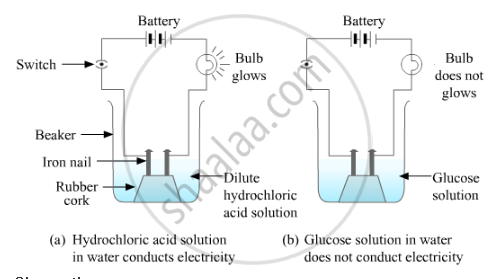
Observation :
The bulb starts glowing in the case of acid, whereas it does not glow in the case of glucose and alcohol solutions. Glowing of the bulb signifies that there is a flow of electric current through the solution. The electric current is carried through the solution by ions.
Conclusion:
Acids dissociate in aqueous solutions to give H+(aq) ions, which determine their acidic property. Glucose and alcohol do not dissociate and do not furnish H+ ions in aqueous solutions even though they contain hydrogen atoms. Hence, HCl shows acidic character in aqueous solutions, whereas solutions of compounds such as C6H12O6 (glucose) and C2H5OH (alcohol) do not show acidic character.
APPEARS IN
संबंधित प्रश्न
On adding dilute hydrochloric acid to copper oxide powder, the solution formed is blue-green.
Predict the new compound formed which imparts a blue-green colour to solution.
What ions are present in the solutions of following substances? (write the symbols only)
Nitric acid
What ions are present in the solutions of following substances? (write the symbols only)
Sulphuric acid
What ions are present in the solutions of following substances? (write the symbols only)
Potassium hydroxide
A first-aid manual suggests that vinegar should be used to treat wasp stings and baking soda for bee stings. What does this information tell you about the chemical nature of:
wasp stings?
Hydrochloric acid reacts with a metal X to form a gas Y which burns with a 'pop' sound. Sodium hydroxide solution also reacts with the same metal X (on heating) to form the same gas Y.
Write the chemical equation of the reaction of metal X with (i) hydrochloric acid, and (ii) sodium hydroxide solution.
State the common and chemical names of the compound formed when plaster of Paris is mixed with water.
During the preparation of hydrogen chloride gas on a humid day, the gas is usually passed through the guard tube containing calcium chloride. The role of calcium chloride taken in the guard tube is to ______
Which of the following gives the correct increasing order of acidic strength?
Some metals react with acids to produce salt and hydrogen gas. Illustrate it with an example. How will you test the presence of this gas?
