Advertisements
Advertisements
प्रश्न
Consider two elements 'X' (Atomic number 17) and 'Y' (Atomic number 20)
(i) Write the positions of these elements in the modern periodic table giving justification.
(ii) Write the formula of the compound formed by the combination of 'X' and 'Y'.
(iii) Draw the electron-dot structure of the compound formed and state the nature of the bond formed between the two elements ?
उत्तर
(i) X belongs to Group 17 and 3rd Period.
Y belongs to Group 2 and 4th Period.
(iii) X is Chlorine while Y is Calcium.
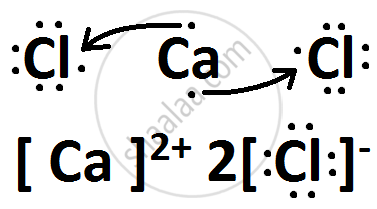
APPEARS IN
संबंधित प्रश्न
In the following set of elements, one element does not belong to the set. Select this element and state why it does not belong:
Oxygen, Nitrogen, Carbon, Chlorine, Fluorine
Name the element which is in first group and third period.
An element which is an essential constituent of all organic compounds belongs to following group of modern periodic table:
(a) group 4
(b) group 14
(c) group 15
(d) group 16
What is its atomic number?
The following questions refer to the Periodic Table.
What is the common feature of the electronic configurations of the elements in group 7?
Choose the most appropriate answer from the following list of oxides which fit the description.
An amphoteric oxide: Al2O3 (shows both acidic and basic properties)
State the nature of compounds formed when group 17 elements combine with (i) metals (ii) non-metals.
The position of certain elements in the Modern Periodic Table is shown below:

Using the above table answer the following questions giving reasons in each case :
(i) Which element will form only covalent compounds?
(ii) Which element is a non-metal with valency 2?
(iii) Which element is a metal with valency 2?
(iv) Out of H, C, and F which has the largest atomic size?
(v) To which family does H, C, and F belong?
Name or state following with reference to the elements of the first three periods of the periodic table.
The number of electron shells in elements of period 1, period 2, and period 3.
Name or state following with reference to the element of the first three periods of the periodic table.
A covalent compound formed between an element in period 1 and a halogen.
