Advertisements
Advertisements
प्रश्न
Consider two elements 'X' (Atomic number 17) and 'Y' (Atomic number 20)
(i) Write the positions of these elements in the modern periodic table giving justification.
(ii) Write the formula of the compound formed by the combination of 'X' and 'Y'.
(iii) Draw the electron-dot structure of the compound formed and state the nature of the bond formed between the two elements ?
उत्तर
(i) X belongs to Group 17 and 3rd Period.
Y belongs to Group 2 and 4th Period.
(iii) X is Chlorine while Y is Calcium.
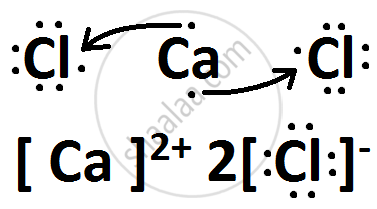
APPEARS IN
संबंधित प्रश्न
Given below are some elements of the modern periodic table. Atomic number of the element is given in parentheses.
A(4), B(9), C(14), D(19), E(20)
(a) Select the element that has one electron in the outermost shell. Also, write the electronic configuration of this element.
(b) Which two elements amongst these belong to the same group? Give reasons for your answer.
(c) Which two elements amongst these belong to the same period? Which one of the two has bigger atomic radius?
How do the atomic size and metallic character of elements vary as we move :-
(a) down a group and
(b) from left to right in a period
The atomic numbers of three elements A, B and C are given below:
| Element | Atomic number |
| A | 5 |
| B | 7 |
| C | 10 |
(i) Which element belongs to group 18?
(ii) Which element belongs to group 15?
(iii) Which element belongs to group 13?
(iv) To which period/periods do these elements belong?
How does the number of:
valence electrons and
Fill in the blank
Oxidising property ________from fluroine to iodine because the power to accpet electron decreases.
Arrange the following as per instruction given in the bracket.
Cl, F, Br, I (increasing electron affinity)
The elements of one short period of the periodic table are given below in order from left to right:
| Li | Be | B | C | O | F | Ne |
To which period do these elements belong?
Moving down in the second group, number of valence electrons ______.
The position of certain elements in the Modern Periodic Table is shown below:

Using the above table answer the following questions giving reasons in each case :
(i) Which element will form only covalent compounds?
(ii) Which element is a non-metal with valency 2?
(iii) Which element is a metal with valency 2?
(iv) Out of H, C, and F which has the largest atomic size?
(v) To which family does H, C, and F belong?
Select the correct answer from the options given below.
It is a period having elements from atomic no. 11 to 18.
