Advertisements
Advertisements
Question
Consider two elements 'X' (Atomic number 17) and 'Y' (Atomic number 20)
(i) Write the positions of these elements in the modern periodic table giving justification.
(ii) Write the formula of the compound formed by the combination of 'X' and 'Y'.
(iii) Draw the electron-dot structure of the compound formed and state the nature of the bond formed between the two elements ?
Solution
(i) X belongs to Group 17 and 3rd Period.
Y belongs to Group 2 and 4th Period.
(iii) X is Chlorine while Y is Calcium.
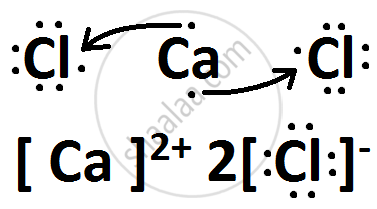
APPEARS IN
RELATED QUESTIONS
Two elements ‘P’ and ‘Q’ belong to the same period of the modern periodic table and are in Group 1 and Group 2, respectively. Compare their following characteristics in tabular form:-
(a) The number of electrons in their atoms
(b) The sizes of their atoms
(c) Their metallic character
(d) Their tendencies to lose electrons
(e) The formula of their oxides
(f) The formula of their chlorides
The electrons in the atoms of four elements A, B, C and D are distributed in three shells having 1, 3, 5 and 7 electrons in the outermost shell respectively. State the period in which these elements can be placed in the modern periodic table. Write the electronic configuration of the atoms of A and D and the molecular formula of the compound formed when A and D combine.
Consider two elements 'A' (Atomic number 17) and 'B' (Atomic number 19) :
(i) Write the positions of these elements in the modern periodic table giving justification.
(ii) Write the formula of the compound formed when 'A' combines with 'B.'
(iii) Draw the electron dot structure of the compound and state the nature of the bond formed between the two elements.
Name two elements you would expect to show chemical reactions similar to calcium. What is the basis of your choice?
Fill in the blank
There are ______ groups and ______ periods in the modern form of periodic table.
Copy and complete the following sentence choosing the correct word or words from those given below, at the end of the sentence:
The similarities in the properties of elements belonging to a group are because they have the same ______
The elements of one short period of the periodic table are given below in order from left to right:
| Li | Be | B | C | O | F | Ne |
To which period do these elements belong?
Name or state following with reference to the elements of the first three periods of the periodic table.
A metalloid in period 2 and in period 3.
Which of the following would weigh the least?
(Atomic masses C = 12, 0 = 16, Na = 23)
There are three elements E, F, G with atomic numbers 19, 8 and 17 respectively.
Classify the above elements as metals and non-metals.
