Advertisements
Advertisements
प्रश्न
Considering x-axis as the molecular axis which out of the following will form a sigma bond.
2px and 2pz
एक शब्द/वाक्यांश उत्तर
उत्तर
2px and 2pz - No sigma bond
shaalaa.com
या प्रश्नात किंवा उत्तरात काही त्रुटी आहे का?
पाठ 10: Chemical bonding - Evaluation [पृष्ठ १०७]
APPEARS IN
संबंधित प्रश्न
Draw an orbital diagram of Fluorine molecule
Give a reason for the sigma (σ) bond is stronger than the Pi (π) bond.
XeF2 is isostructural with ______.
Among the following, the compound that contains, ionic, covalent and Coordinate linkage is ______.
Define σ – bond.
What is a pi - bond?
Considering x-axis as the molecular axis which out of the following will form a sigma bond.
2px and 2py
The number of sigma bonds in vanillin is ____________.
Why does type of overlap given in the following figure not result in bond formation?
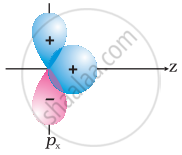 |
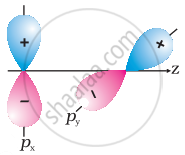 |
The \[\ce{H - N - H}\] bond angle in ammonia molecule is ______.
