Advertisements
Advertisements
प्रश्न
Considering x-axis as the molecular axis which out of the following will form a sigma bond.
1s and 2pz
उत्तर
1s and 2pz - No sigma bond
APPEARS IN
संबंधित प्रश्न
Draw diagram for bonding in ethene with sp2 Hybridisation.
Draw an orbital diagram of Fluorine molecule
Give a reason for carbon is tetravalent in nature.
Identify the type of overlap present in F2. Explain diagrammatically.
Give the type of overlap by which the pi (π) bond is formed.
Define σ – bond.
Considering x-axis as the molecular axis which out of the following will form a sigma bond.
2px and 2py
If the electronic configuration of an element is 1s2 2s2 2p6 3s2 3p6 3d2 4s2, the four electrons involved in chemical bond formation will be ______.
Why does type of overlap given in the following figure not result in bond formation?
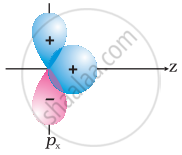 |
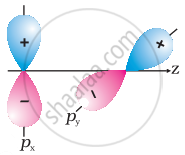 |
Briefly describe the valence bond theory of covalent bond formation by taking an example of hydrogen. How can you interpret energy changes taking place in the formation of dihydrogen?
