Advertisements
Advertisements
प्रश्न
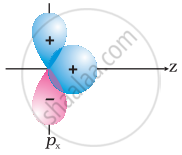
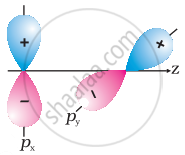
Why does type of overlap given in the following figure not result in bond formation?
 |
 |
उत्तर
In the first figure given above, the area which is under ++ overlapping is equal to the area under +– overlap. Both the overlaps cancel out with each other as they are oppositely charged. Due to cancelling out of the overlaps the net overlap will be zero.
In the second figure given above, both the p-orbitals are perpendicular to each other. Due to the `p_x, p_y` orbitals being perpendicular with each other, no overlap will be possible.
APPEARS IN
संबंधित प्रश्न
Distinguish between sigma and pi bond.
Identify the type of overlap present in H2. Explain diagrammatically.
Which one of the following is the likely bond angles of sulphur tetrafluoride molecule?
In ClF3, NF3 and BF3 molecules the chlorine, nitrogen and boron atoms are ______.
When ones and three p orbitals hybridise,
Which of these represents the correct order of their increasing bond order.
The correct order of O – O bond length in hydrogen peroxide, ozone and oxygen is
XeF2 is isostructural with ______.
Considering x-axis as the molecular axis which out of the following will form a sigma bond.
2px and 2py
Ethene molecule has ____________ sp2 -s σ bond(s), ____________ sp2 -sp2 σ bond(s) and ____________ p-p π bond(s).
