Advertisements
Advertisements
प्रश्न
Which one of the following is the likely bond angles of sulphur tetrafluoride molecule?
पर्याय
120°, 80°
109°28’
90°
89°, 117°
उत्तर
89°, 117°
APPEARS IN
संबंधित प्रश्न
Distinguish between sigma and pi bond.
Give reasons for need of Hybridization
Identify the type of overlap present in F2. Explain diagrammatically.
When ones and three p orbitals hybridise,
Which bond is stronger σ or π? Why?
Considering x-axis as the molecular axis which out of the following will form a sigma bond.
2px and 2py
Considering x-axis as the molecular axis which out of the following will form a sigma bond.
1s and 2pz
The number of sigma bonds in vanillin is ____________.
Why does type of overlap given in the following figure not result in bond formation?
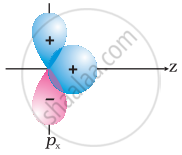 |
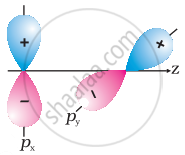 |
Match List - I with List - II.
| List - I | List - II | ||
| (a) | \[\ce{PCl5}\] | (i) | Square pyramidal |
| (b) | \[\ce{SF6}\] | (ii) | Trigonal planar |
| (c) | \[\ce{BrF5}\] | (iii) | Octahedral |
| (d) | \[\ce{BF3}\] | (iv) | Trigonal bipyramidal |
Choose the correct answer from the options given below.
