Advertisements
Advertisements
प्रश्न
The ratio of number of sigma (σ) and pi (л) bonds in 2- butynal is ______.
पर्याय
`8/3`
`5/3`
`8/2`
`9/2`
उत्तर
The ratio of number of sigma (σ) and pi (л) bonds in 2- butynal is `underline(8/3)`.
APPEARS IN
संबंधित प्रश्न
Complete the following Table.
| Molecule | Type of Hybridization | Type of bonds | Geometry | Bond angle |
| CH4 | - | 4C-H 4σ bonds |
Tetrahedral | - |
| NH3 | sp3 | 3N-H 3σ bonds 1 lone pair |
- | - |
| H2O | - | - | angular | 104.5° |
| BF3 | sp2 | - | - | 120° |
| C2H4 | - | - | - | 120° |
| BeF2 | - | 2 Be-F | Linear | - |
| C2H2 | sp | (3σ+2π) 1C-C σ 2C-H σ 2C-C π |
- | - |
Mention the steps involved in Hybridization.
The correct order of O – O bond length in hydrogen peroxide, ozone and oxygen is
XeF2 is isostructural with ______.
What is a pi - bond?
Which bond is stronger σ or π? Why?
Considering x-axis as the molecular axis which out of the following will form a sigma bond.
1s and 2py
Considering x-axis as the molecular axis which out of the following will form a sigma bond.
1s and 2pz
If the electronic configuration of an element is 1s2 2s2 2p6 3s2 3p6 3d2 4s2, the four electrons involved in chemical bond formation will be ______.
Why does type of overlap given in the following figure not result in bond formation?
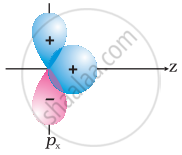 |
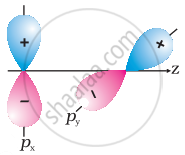 |
