Advertisements
Advertisements
प्रश्न
Chlorine, nitrogen, ammonia and sulphur dioxide gases are collected under the same conditions of temperature and pressure.
Copy the following table which gives the volumes of the gases collected, and the number of molecules (X) in 20L of nitrogen.You are to complete the table by giving the number of molecules in th e other gases, in terms of X.
| Gas | Volume(litres) | Number of molecules |
| Chlorine | 10 | |
| Nitrogen | 20 | X |
| Ammonia | 20 | |
| Sulphur dioxide | 5 |
उत्तर
(a) If 20L of nitrogen has = X number of molecules
Then, 10L of chlorine will have = X x 10/20 = X/2.
(b) If 20L of nitrogen has = X number of molecules
Then, 20L of ammonia will have = X x 20/20 = X.
(c) If 20L of nitrogen has = X number of molecules
Then, 5L of sulphur dioxide will have = X x 5/20 = X/4.
| Gas | Volume(litres) | Number of molecules |
| Chlorine | 10 | X/2 |
| Nitrogen | 20 | X |
| Ammonia | 20 | X |
| Sulphur dioxide | 5 | X/4 |
APPEARS IN
संबंधित प्रश्न
Give balanced chemical equations for the following conversions A, B, and C:
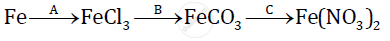
The usefulness of a fertilizer depends upon percentage of nitrogen present in it. Find which of the following is a better fertilizer:
(a) Ammonium nitrate [NH4NO3]
(b) Ammonium phosphate [(NH4)3PO4 (N=14,H=1,O=16,P=31)
Potassium nitrate on strong heating decomposes as under :
2KNO3 → 2KNO2 + O2
Calculate : Weight of oxygen formed when 5.05g of potassium nitrate decomposes completely.
(K = 39, 0 = 16, N = 14)
Calculate the percentage of water in ferrous sulphate crystals.
[Fe = 56, S = 32, O =16, H = 1].
Calculate the percentage of sodium in sodium aluminium fluoride (Na3AIF6).
[F = 19, Na = 23, Al = 27]
A gaseous hydrocarbon contains 82.76% of carbon. Given that its vapour density is 29, find its molecular formula.
[C = 12, H = 1]
Give two tests of the following:
Oxygen
Calculate the relative molecular mass of:
Ammonium chloroplatinate (NH4)2 PtCl6
A gas cylinder can hold 1 kg of hydrogen at room temperature and pressure. What mass of carbon dioxide can it hold under similar conditions of temperature and pressure?
Ammonia burns in oxygen and the combustion, in the presence of a catalyst, may be represented by;
\[\ce{2NH3 + 2 1/2O2 -> 2NO + 3H2O}\] [H = 1, N = 14, O = 16]
What mass of steam is produced when 1.5 g of nitrogen monoxide is formed?
