Advertisements
Advertisements
प्रश्न
Define: Endothermic reaction
उत्तर
The reactions in which heat is absorbed are called endothermic reactions. The reactants absorb heat for form products.
APPEARS IN
संबंधित प्रश्न
Balance the chemical equation.
\[\ce{HNO3 +Ca(OH)2 -> Ca(NO3)2 + H2O}\]
Balance the following chemical equation.
\[\ce{NaCl + AgNO3 -> AgCl + NaNO3}\]
Write the balanced chemical equation for the following reaction.
\[\ce{Aluminium + Copper chloride -> Aluminium chloride + Copper}\]
Complete and balance the following equation:
NaOH + ............  Na2 SO4 + H2O
Na2 SO4 + H2O
Balance the given equation:
AI(OH)3  AI2O3 +H2O
AI2O3 +H2O
State one characteristic of the chemical reaction which takes place when lemon juice is added gradually to potassium permanganate solution.
An acid which can decolourise purple coloured potassium permanganate solution is:
(a) sulphuric acid
(b) citric acid
(c) carbonic acid
(d) hydrochloric acid
When water is added gradually to a white solid X, a hissing sound is heard and a lot of heat is produced forming a product Y. A suspension of Y in water is applied to the walls of a house during white washing. A clear solution of Y is also used for testing carbon dioxide gas in the laboratory.
(a) What could be solid X? Write its chemical formula.
(b) What could be product Y? Write its chemical formula.
(c) What is the common name of the solution of Y which is used for testing carbon dioxide gas?
(d) Write chemical equation of the reaction which takes place on adding water to slid X.
(e) Which characteristic of chemical reactions is illustrated by this example?
A metal X forms a salt XSO4. The salt XSO4 forms a clear solution in water which reacts with sodium hydroxide solution to form a blue precipitate Y. Metal X is used in making electric wire and alloys like brass.
(a) What do you think metal X could be?
(b) Write the name, formula and colour of salt XSO4.
(c) What is the blue precipitate Y?
(d) Write a chemical equation of the reaction which takes place when salt XSO4 reacts with sodium hydroxide solution. Give the state symbols of all the reactants and products which occur in the above equation.
Write balanced chemical equation with state symbols for the following reaction:
Barium chloride solution reacts with sodium sulphate solution to give insoluble barium sulphate and a solution of sodium chloride.
What is a chemical equation?
Write word equation for the following skeletal equation:
\[\ce{KClO3 -> KCl + O2}\]
Balance the equation stepwise.
Ag(s) + HCl(aq) → AgCl ↓+ H2 ↑
Balance the following equation:
Pb3O4 → PbO + O2
Balance the following equation:
H2O + Cl2 → HCl + O2
Why should an equation be balanced? Explain with the help of a simple equation.
Write the balanced chemical equation of the following reaction.
aluminium carbide + water → aluminium hydroxide + methane
Write the balanced chemical equation of the following reaction.
Potassium permanganate + hydrochloric acid → potassium chloride + manganese chloride + chlorine + water
Write the balanced chemical equation of the following reaction.
sulphur + nitric acid→ sulphuric acid + nitrogen dioxide + water.
Write the balanced chemical equation of the following reaction.
sodium chloride + manganese dioxide + sulphuric acid → sodium hydrogen sulphate + manganese sulphate + water + chlorine.
\[\ce{MnO2 + 4HCl -> MnCl2 + 2H2O + Cl2}\]
0.02 moles of pure MnO2 is heated strongly with conc. HCl. Calculate the mass of salt formed.
A student mixes sodium sulphate powder in barium chloride powder. What change would the student observe on mixing the two powders? Justify your answer and explain how he can obtain the desired change.
Write word equation for the following molecular equation:
Na2SO3 + 2HCl [dil.] → 2NaCl + H2O + SO2 [g]
Write word equation for the following molecular equation:
CaO + H20 → Ca (OH)2 + Δ
Word equation:
State why [+ Δ ] is not written in the reaction, if ZnO reacts with H2O.
Write word equation for the following molecular equation:
\[\ce{CuSO4 + 2NaOH -> Na2SO4 + Cu(OH)2↓}\]
Word equation:
State the colour of the products.
Give word equation for the following chemical reaction and give the names of the product formed.
NH3 + HCl→ NH4Cl
Write word equation for the following chemical reaction given below. Also state the observation seen in the case.
\[\ce{ZnCo3->[\triangle] ZnO + CO2}\]
Write word equation for the following chemical reaction given below. Also state the observation seen in the case.
\[\ce{2SO2 + O2 ⇌[V2O5][450°C] 2SO3}\]
Balance the following simple equation:
ZnS + O2 → ZnO + SO2
Balance the following simple equation:
SO2 + O2 ⇌ SO3
Balance the following simple equation:
\[\ce{Ca(OH)2 + HNO3 -> Ca(NO3)2 + H2O}\]
Balance the following simple equation:
Al + H2SO4 → Al2(SO4)3 + H2
Balance the following simple equation:
NaHCO3 + H2SO4 → Na2SO4 + H2O + CO2
Write a balanced equation for the following word equation:
Aluminium + Oxygen → Aluminium oxide
Write a balanced equation for the following word equation:
Calcium hydroxide + Ammonium chloride → Calcium chloride + Water + Ammonia
Give a balanced equation by the partial equation method, [steps are given below].
Oxidation of Lead [II] Sulphide by Ozone
- Ozone first decomposes to give molecular oxygen & nascent oxygen.
- Nascent oxygen then oxidises lead [II] sulphide to lead [II] sulphate.
Balance the given chemical reaction as per the instructions below.
\[\ce{NaOH + H2SO4 -> Na2SO4 + H2O}\]
Balance the equation with proper coefficient and rewrite the equation.
Which among the following statement(s) is(are) true? ‘Exposure of silver chloride to sunlight for a long duration turns grey due to
- the formation of silver by decomposition of silver chloride
- sublimation of silver chloride
- decomposition of chlorine gas from silver chloride
- oxidation of silver chloride
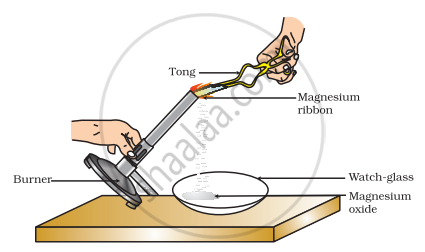
Which of the following is the correct observation of the reaction shown in the above set up?
