Advertisements
Advertisements
प्रश्न
Discuss the nature of C – X bond in the haloarenes.
उत्तर
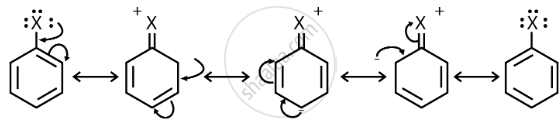
Resonance effect: Because the C – X bond has the characteristics of a partial double bond, it is extremely difficult to break.
(i) The C – X bond in haloarenes is extremely less reactive to nucleophilic groups.
(ii) The C atom linked to the halogen is sp2 hybridised in the C – X bond. Carbon that has been sp2 hybridised with a higher s-character is more electronegative in nature and can retain the electron pair of the C – X bond more tightly than carbon that has been sp3 hybridised with a lower s-character in haloalkanes.
APPEARS IN
संबंधित प्रश्न
which of the following carbocations is least stable?
(A) 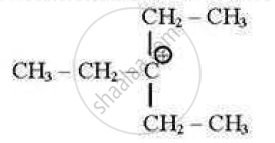
(B)![]()
(C)![]()
(D)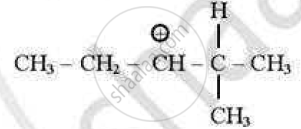
Which one of the following has the highest dipole moment?
C – X bond is strongest in ____________.
Which of the following will have the maximum dipole moment?
Write a test to detect the presence of double bond in a molecule.
Assertion: Nitration of chlorobenzene leads to the formation of m-nitrochlorobenzene.
Reason: –NO2 group is a m-directing group.
Out of the Chloromethane and Fluoromethane, which one is has higher dipole moment and why?
Why haloarenes are Less reactive than halo alkanes?
