Advertisements
Advertisements
प्रश्न
Write a test to detect the presence of double bond in a molecule.
उत्तर
The Presence of a double bond in a molecule can be detected by bromine water test and Baeyer’s test. The molecules with a double bond decolourise bromine water and aqueous \[\ce{KMnO4}\].
\[\begin{array}{cc}
\phantom{}\ce{> C = C < + \underset{Brown}{Br2} -> > C - C <}\phantom{}\\
\phantom{.........................}|\phantom{.....}|\phantom{}\\
\phantom{...........................}\ce{\underset{Colourless}{\phantom{.}Br\phantom{...}Br}}\phantom{..}
\end{array}\]
\[\begin{array}{cc}
\phantom{...}\ce{> C = C < ->[aq. KMnO4][(Purple)] > C - C <}\phantom{}\\
\phantom{..........................}|\phantom{....}|\phantom{}\\
\phantom{..............................}\ce{\underset{Colourless}{\phantom{.}OH\phantom{.}OH}}\phantom{...}
\end{array}\]
APPEARS IN
संबंधित प्रश्न
which of the following carbocations is least stable?
(A) 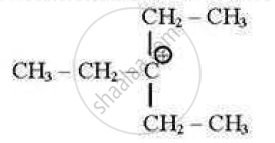
(B)![]()
(C)![]()
(D)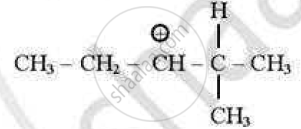
Which one of the following has the highest dipole moment?
C – X bond is strongest in ____________.
Which of the following will have the maximum dipole moment?
Discuss the nature of C – X bond in the haloarenes.
Assertion: Nitration of chlorobenzene leads to the formation of m-nitrochlorobenzene.
Reason: –NO2 group is a m-directing group.
Out of the Chloromethane and Fluoromethane, which one is has higher dipole moment and why?
Why haloarenes are Less reactive than halo alkanes?
