Advertisements
Advertisements
प्रश्न
How do polar solvents help in the first step in SN1 mechanism?
उत्तर
The SN1 mechanism proceeds through the formation of carbocation. It involves breaking of C-halogen bond for which energy is obtained through the salvation of halide ion with the proton of the protic solvent. Thus, polar solvents help in ionisations step by stabilizing the ions by solvation.
APPEARS IN
संबंधित प्रश्न
How do you convert the following:
Ethanol to propanenitrile
In the following pair of halogen compounds, which compound undergoes a faster SN1 reaction?
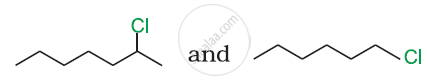
What happens when methyl chloride is treated with KCN?
C–Cl bond length in chlorobenzene is shorter than C–Cl bond length in CH3–Cl.
Which would undergo SN2 reaction faster in the following pair and why ?
CH3 – CH2 – Br and CH3 – CH2 – I
A primary alkyl halide would prefer to undergo ______.
Write the structures and names of the compounds formed when compound ‘A’ with molecular formula, \[\ce{C7H8}\] is treated with \[\ce{Cl2}\] in the presence of \[\ce{FeCl3}\].
Cyanide ion acts as an ambident nucleophile. From which end it acts as a stronger nucleophile in aqueous medium? Give reason for your answer.
Which one is the correct order of nucleophilic strength (pKa) of following nucleophiles?
Give the mechanism of the following reaction:
\[\ce{CH3CH2OH ->[H2SO4][413 K] CH3CH2-O-CH2CH3 + H2O}\]
