Advertisements
Advertisements
प्रश्न
Why is it necessary to avoid even traces of moisture during the use of a Grignard reagent?
उत्तर
Grignard reagents are highly reactive and react with even traces of water to give corresponding hydrocarbons.
\[\ce{RMgX + H2O –>RH + Mg(OH)X}\]
APPEARS IN
संबंधित प्रश्न
Explain why Grignard reagents should be prepared under anhydrous conditions?
Write the structure of the major organic product in the following reaction:
\[\ce{C6H5ONa + C2H5Cl ->}\]
Write the structure of main compounds A and B in the following reaction: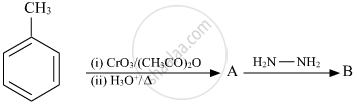
A Grignard reagent may be made by reacting magnesium with ____________.
Draw other resonance structures related to the following structure and find out whether the functional group present in the molecule is ortho, para directing or meta directing.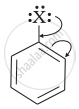
Reaction of Grignard reagent with aromatic aldehyde and subsequent aqueous treatment produces
\[\ce{R - CH2 - CCl2 - R ->[Peagent] R - C ≅ C - R}\]. The reagent is:-
Explain why Grignard reagents should be prepared under anhydrous conditions?
Explain why Grignard reagents should be prepared under anhydrous conditions?
Explain why Grignard reagents should be prepared under anhydrous conditions?
