Advertisements
Advertisements
प्रश्न
Write the structure of the major organic product in the following reaction:
\[\ce{C6H5ONa + C2H5Cl ->}\]
उत्तर १
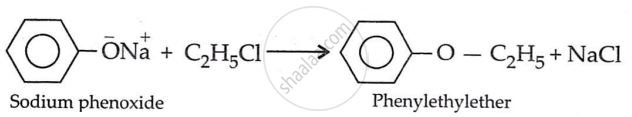
\[\ce{\underset{sodium phenoxide}{C6H5O^-Na} + \underset{ethyl chloride}{C2H5Cl} ->[Williamson Synthesis] \underset{phenetole}{C6H5 - O - C2H5} + NaCl}\]
उत्तर २
Notes
Students can refer to the provided solutions based on their preferred marks.
APPEARS IN
संबंधित प्रश्न
How will you bring about the following conversion?
Bromomethane to propanone
How will you bring about the following conversion?
1-Chlorobutane to n-octane
What happens when bromobenzene is treated with Mg in the presence of dry ether?
How the following conversion can be carried out?
2-Chlorobutane to 3, 4-dimethylhexane
How the following conversion can be carried out?
Chloroethane to butane
A Grignard reagent may be made by reacting magnesium with ____________.
Which one of the following produces acyl halide by treatment with PCl5?
What would be the reactant and reagent used to obtain 2, 4-dimethyl pentan-3-ol?
Which of the following statements are correct about the reaction intermediate?
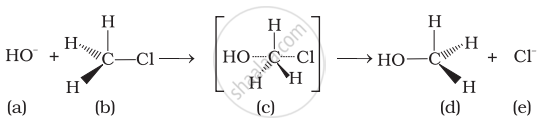
(i) Intermediate (c) is unstable because in this carbon is attached to 5 atoms.
(ii) Intermediate (c) is unstable because carbon atom is sp2 hybridised.
(iii) Intermediate (c) is stable because carbon atom is sp2 hybridised.
(iv) Intermediate (c) is less stable than the reactant (b).
Why is it necessary to avoid even traces of moisture during the use of a Grignard reagent?
\[\ce{R - CH2 - CCl2 - R ->[Peagent] R - C ≅ C - R}\]. The reagent is:-
Grignard reagent shows addition on
Explain why Grignard reagents should be prepared under anhydrous conditions?
Explain why Grignard reagents should be prepared under anhydrous conditions?
Explain why Grignard reagents should be prepared under anhydrous conditions?
Explain why Grignard reagents should be prepared under anhydrous conditions?
Explain why Grignard reagents should be prepared under anhydrous conditions?
