Advertisements
Advertisements
प्रश्न
Dissolving salt in water is a reversible change.
उत्तर
Dissolving salt in water is a reversible change because salt and water can be again obtained by distillation process.
APPEARS IN
संबंधित प्रश्न
Convert the following temperature to the celsius scale.
470 K
Name A, B, C, D, E and F in the following diagram showing change in its state.
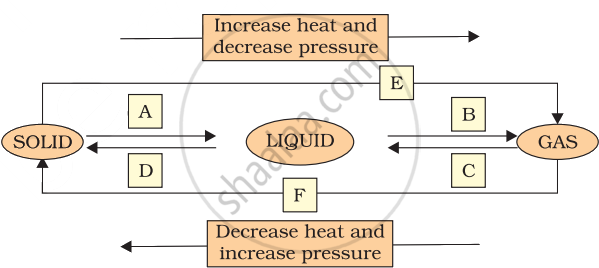
What do you mean by the change of state? Write the flow chart showing the complete cycle of change of state.
Mention a change which is always desirable.
Tick (√) the correct alternative from the choice given for the following statement.
When we add water to the following substances, which one will show a chemical change?
What do you understand by the change of phase of a substance?
A few substances are arranged in the increasing order of 'forces of attraction' between their particles. Which one of the following represents the correct arrangement?
Convert the temperature of 373° C to the Kelvin scale.
Explain why, there is no rise in temperature of a substance when it undergoes a change of state though heat is supplied continuously.
One of the following does not undergo sublimation. This one is :
Define the following terms: Sublimation
Differentiate between: Solidification and condensation
Give reasons:
Why a solid does not flow, but a liquid flows?
How does a liquid changes into its gaseous state? Explain
Name the phenomenon which causes the following changes:
Conversion of ice into water
Give two examples of the following:
Substances which do not change their states.
The process by which a solid changes into a liquid is called
State which of the three states of matter Le. solids, liquids or gases – have
(a) No definite volume
(b) A definite shape
(c) High density
(d) No free surfaces
(e) Particles – which diffuse very easily.
Give the reason for the following
On heating a sublimable solid, the molecules break free & escape from the surface of the solid directly into vapour.
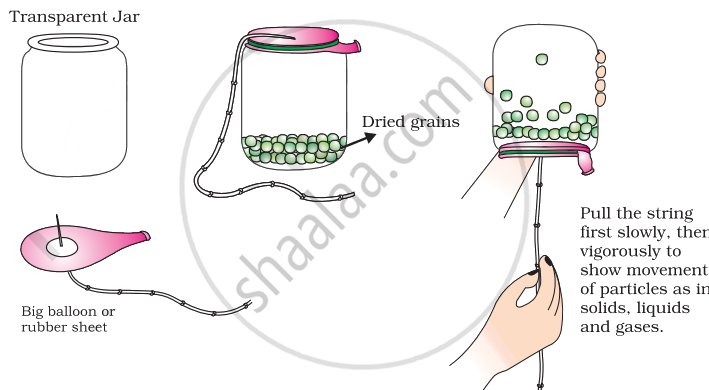
Prepare a model to demonstrate movement of particles in solids, liquids and gases.
For making this model you will need
- A transparent jar
- A big rubber balloon or piece of stretchable rubber sheet
- A string
- Few chickpeas or black gram or dry green peas.
How to make?
- Put the seeds in the jar.
- Sew the string to the centre of the rubber sheet and put some tape to keep it tied securely.
- Stretch and tie the rubber sheet on the mouth of the jar.
- Your model is ready. Now run your fingers up and down the string by first tugging at it slowly and then rapidly.