Advertisements
Advertisements
प्रश्न
Explain why, there is no rise in temperature of a substance when it undergoes a change of state though heat is supplied continuously.
उत्तर
There is no rise in the temperature of a substance when it undergoes a change of state though heat is being supplied because heat is used to overcome inter-molecular force of attraction between the particles of the substance during a change in physical state.
APPEARS IN
संबंधित प्रश्न
Convert the following temperature to Celsius scale:
300 K
What is the physical state of water at 100°C.
The latent heat of fusion of ice is :
Define: cohesive force
Define: Brownian movement
Define the following terms: Vaporisation
Name the phenomenon which causes the following changes:
Formation of water vapour from water
What will happen if....? And why?
Rubber is not fitted on a pair of tongs.
Why does this happen?
Kerosene left open in a dish disappears.
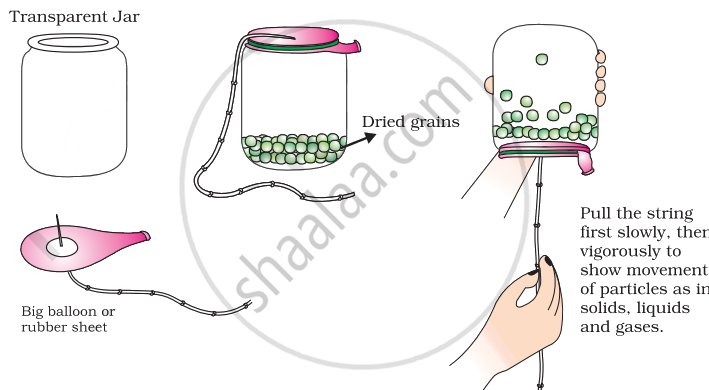
Prepare a model to demonstrate movement of particles in solids, liquids and gases.
For making this model you will need
- A transparent jar
- A big rubber balloon or piece of stretchable rubber sheet
- A string
- Few chickpeas or black gram or dry green peas.
How to make?
- Put the seeds in the jar.
- Sew the string to the centre of the rubber sheet and put some tape to keep it tied securely.
- Stretch and tie the rubber sheet on the mouth of the jar.
- Your model is ready. Now run your fingers up and down the string by first tugging at it slowly and then rapidly.