Advertisements
Advertisements
प्रश्न
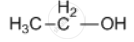
Draw the structural formula of a compound with two carbon atoms in the following case.
An alcohol containing two carbon atoms
उत्तर
APPEARS IN
संबंधित प्रश्न
Give an example of isomers shown by triple bond hydrocarbons (alkynes) and write their IUPAC names.
Complete and balance the following equation. State the condition wherever necessary.
C2H4 + Br2 → ______
Complete and balance the following equation. State the condition wherever necessary.
\[\ce{C2H5OH ->[O][K2Cr2O7]}\] ______
Explain the following:
Tetravalency
Give at least one example in case to show the structure of isomers of a triple bond compound.
Define the substitution reaction.
What will be the formula and structure of benzene?
Fill in the blanks with the correct words from the brackets:
Alkenes are the (i) ______ (analogous/homologous) series of (ii) ______ (saturated/unsaturated) hydrocarbons. They differ from alkanes due to presence of (c) ______ (double/single) bonds. Alkenes mainly undergo (iv) ______ (addition/substitution) reactions.
Name an alcohol with three carbon atom.
The structures of four hydrocarbons are shown below:
| \[\begin{array}{cc} \phantom{.....}\ce{CH3}\\ \phantom{...}|\\ \ce{H3C - C - H}\\ \phantom{...}|\\ \phantom{.....}\ce{CH3} \end{array}\] |
\[\begin{array}{cc} \ce{CH3}\phantom{.....}\\ |\phantom{.......}\\ \ce{C = CH2}\\ |\phantom{.......}\\ \ce{CH3}\phantom{....} \end{array}\] |
\[\begin{array}{cc} \ce{H}\phantom{.....}\\ |\phantom{.....}\\ \ce{H3C - C - C - CH2}\\ |\phantom{....}|\\ \ce{H\phantom{...}H} \end{array}\] |
\[\begin{array}{cc} \phantom{...}\ce{CH3}\\ |\\ \ce{H3C - C = CH2} \end{array}\] |
How many isomers of butene are there?
