Advertisements
Advertisements
प्रश्न
Draw the structure of the following atoms showing the nucleus containing – protons, neutrons and the orbits with the respective electron:
Carbon [At. no. = 6, Mass no. = 12]
उत्तर
Structure of atoms:
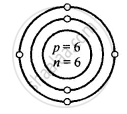
Z is Atomic Number and A is the mass number
Carbon \[\ce{^12_6C}\]
Z = 6 = p = e
K L
e = 6 = 2, 4
A = p + n
12 = 6 + n
∴ n = 12 − 6 = 6
APPEARS IN
संबंधित प्रश्न
The composition of the nuclei of two atomic species X and Y are given as under
| X | Y | |
| Protons | 6 | 6 |
| Neutrons | 6 | 8 |
Give the mass numbers of X and Y. What is the relation between the two species?
FILL IN THE BLANK
Rutherford won the Nobel Prize in 1908 for his investigations in the field of ......................
The atomic number of four elements A, B, C and D are 12, 13, 15 and 3 respectively. The element which cannot form a cation is :
The properties of the elements are a periodic function of their __________.
Elements X, Y and Z have atomic numbers 6, 9 and 12 respectively. Which one:
- Forms an anion
- Forms a cation
- Has four electrons in its valence shell?
The diagram represents an isotope of hydrogen [H]. Answer the following:

Atomic no. = 1
Mass no. = 1
If an isotope of ‘H’ has mass no. = 3, how many neutrons does it have.
The diagram represents an isotope of hydrogen [H]. Answer the following:

Atomic no. = 1
Mass no. = 1
Which sub-atomic particles in the 3 isotopes of ‘H’ are the same.
Differentiate between the following term:
Atomic number and mass number
Ca2+ has completely filled outer shell. Justify your answer.
A group of elements in the periodic table is given below (Boron is the first member of the group and Thallium is the last).
Boron, Aluminium, Gallium, Indium, Thallium
Answer the following question in relation to the above group of elements:
The atomic number of boron is 5. Write the chemical formula of the compound formed when boron reacts with chlorine.
