Advertisements
Advertisements
प्रश्न
Draw the structure of the following atoms showing the nucleus containing – protons, neutrons and the orbits with the respective electron:
Isotopes of hydrogen [11H, 21H, 31H]
उत्तर
Structure of atoms:

\[\ce{^2_1H}\]

\[\ce{^3_1H}\]
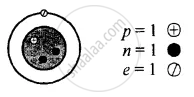
Z is Atomic Number and A is the mass number
Isotopes of hydrogen \[\ce{^1_1H}\]
Z = 1 = p = e
∴ e = 1
A = p + n
1 = 1 + n
∴ n = 1 − 1 = 0
APPEARS IN
संबंधित प्रश्न
With the help of given Table, find out the mass number of sulphur atom.
| Composition of Atoms of the First Eighteen Elements with Electron Distribution in Various Shells | ||||||||||
| Name of Element | Symbol | Atomic Number | Number of protons | Number of Neutrons | Number of Electrons | Distribution of Electrons | Valency | |||
| K | L | M | N | |||||||
| Hydrogen | H | 1 | 1 | - | 1 | 1 | - | - | - | 1 |
| Helium | He | 2 | 2 | 2 | 2 | 2 | - | - | - | 0 |
| Lithium | Li | 3 | 3 | 4 | 3 | 2 | 1 | - | - | 1 |
| Beryllium | Be | 4 | 4 | 5 | 4 | 2 | 2 | - | - | 2 |
| Boron | B | 5 | 5 | 6 | 5 | 2 | 3 | - | - | 3 |
| Carbon | C | 6 | 6 | 6 | 6 | 2 | 4 | - | - | 4 |
| Nitrogen | N | 7 | 7 | 7 | 7 | 2 | 5 | - | - | 3 |
| Oxygen | O | 8 | 8 | 8 | 8 | 2 | 6 | - | - | 2 |
| Fluorine | F | 9 | 9 | 10 | 9 | 2 | 7 | - | - | 1 |
| Neon | Ne | 10 | 10 | 10 | 10 | 2 | 8 | - | - | 0 |
| Sodium | Na | 11 | 11 | 12 | 11 | 2 | 8 | 1 | - | 1 |
| Magnesium | Mg | 12 | 12 | 12 | 12 | 2 | 8 | 2 | - | 2 |
| Aluminium | Al | 13 | 13 | 14 | 13 | 2 | 8 | 3 | - | 3 |
| Silicon | Si | 14 | 14 | 14 | 14 | 2 | 8 | 4 | - | 4 |
| Phosphorus | P | 15 | 15 | 16 | 15 | 2 | 8 | 5 | - | 3.5 |
| Sulphur | S | 16 | 16 | 16 | 16 | 2 | 8 | 6 | - | 2 |
| Chlorine | Cl | 17 | 17 | 18 | 17 | 2 | 8 | 7 | - | 1 |
| Argon | Ar | 18 | 18 | 22 | 18 | 2 | 8 | 8 | 0 | |
What are the atomic numbers of elements whose outermost electrons is represented by 3p5?
Multiple Choice Questions
The sub atomic particle with negative charge is
Fill in the blank of the following statement :
All the mass of an atom is concentrated in a small region of space called the ___________.
An element A atomic number 7 mass numbers 14
B electronic configuration 2,8,8
C electrons 13, neutrons 14
D Protons 18 neutrons 22
E Electronic configuration 2,8,8,1
State Valency of each element
An element A atomic number 7 mass numbers 14
B electronic configuration 2,8,8
C electrons 13, neutrons 14
D Protons 18 neutrons 22
E Electronic configuration 2,8,8,1
State (i) Valency of each element (ii) which one is a metal (iii) which is non-metal (iv) which is an inert gas
Define the term ‘atomic number’ of an atom.
Draw the atomic diagram of the following element showing the distribution of – protons, neutrons and the electrons in the various shell of the atom.
Calcium – \[\ce{^40_20Ca}\]
[The upper number represent the – mass number and the lower number represent the – atomic number e.g. calcium – mass number = 40, atomic number = 20]
The diagram represents an isotope of hydrogen [H]. Answer the following:

Atomic no. = 1
Mass no. = 1
If an isotope of ‘H’ has mass no. = 2, how many electrons does it have.
Atomic number : ______ :: Mass number : Number of protons and neutrons.
The atomic number and the mass number of an element is 26 and 56 respectively. Calculate the number of electrons, protons and neutrons in its atom. Draw the structure.
