Advertisements
Advertisements
प्रश्न
Draw the structure of the following atoms showing the nucleus containing – protons, neutrons and the orbits with the respective electron:
Isotopes of hydrogen [11H, 21H, 31H]
उत्तर
Structure of atoms:

\[\ce{^2_1H}\]

\[\ce{^3_1H}\]
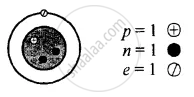
Z is Atomic Number and A is the mass number
Isotopes of hydrogen \[\ce{^1_1H}\]
Z = 1 = p = e
∴ e = 1
A = p + n
1 = 1 + n
∴ n = 1 − 1 = 0
APPEARS IN
संबंधित प्रश्न
Draw the orbital diagram of `""_20^40"Ca"^(2+)` ion and state the number of three fundamental particles present in it.
From the symbol `""_2^4"He"` for the element helium, write down the mass number and the atomic number of the element.
Match the statements in List I with the correct answer from List II.
| List I | List II |
| 1. Mass number of an atom is the number of protons and | A: Electron |
| 2. The sub-atomic particle with a negligible mass. | B: Argon |
| 3. An atom having stable electronic configuration. | C: Nitrogen |
| 4. A molecule formed by sharing of electrons [covalency] | D: Sodium |
| 5. A metallic atom having unstable electronic configuration. | E: Neutrons |
The diagram represents an isotope of hydrogen [H]. Answer the following:

Atomic no. = 1
Mass no. = 1
Do isotopes have the same atomic number or the same mass number?
The diagram represents an isotope of hydrogen [H]. Answer the following:

Atomic no. = 1
Mass no. = 1
Which sub-atomic particles in the 3 isotopes of ‘H’ are the same.
Draw the structure of the following atoms showing the nucleus containing – protons, neutrons and the orbits with the respective electron:
Lithium [At. no. = 3, Mass no. = 7]
Which of the following are true for an element?
- Atomic number = number of protons + number of electrons
- Mass number = number of protons + number of neutrons
- Atomic mass = number of protons = number of neutrons
- Atomic number = number of protons = number of electrons
The Atomic number of an element is 20. It has ______ valence electrons.
Electrons revolve around the nucleus in definite open shells.
K and L shells can accommodate a maximum of 2 and 8 electrons respectively.
