Advertisements
Advertisements
Question
Draw the structure of the following atoms showing the nucleus containing – protons, neutrons and the orbits with the respective electron:
Isotopes of hydrogen [11H, 21H, 31H]
Solution
Structure of atoms:

\[\ce{^2_1H}\]

\[\ce{^3_1H}\]
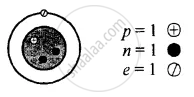
Z is Atomic Number and A is the mass number
Isotopes of hydrogen \[\ce{^1_1H}\]
Z = 1 = p = e
∴ e = 1
A = p + n
1 = 1 + n
∴ n = 1 − 1 = 0
APPEARS IN
RELATED QUESTIONS
FILL IN THE BLANK
Atomic theory of matter was given by ..........................
ÅIn group I of the periodic table, three elements X, Y, and Z have ionic radii 1.33 Å, 0.95 Å and 0.60 Å respectively. Giving a reason, arrange them in the order of increasing atomic number in the group.
The number of electrons in the atom of an element X is 15 and the number of neutrons is 16. Which of the following is the correct representation of an atom of this element ?
From the symbol `""_15^31"P"`, state : atomic number of phosphorus
Draw the orbital diagram of `""_20^40"Ca"^(2+)` ion and state the number of three fundamental particles present in it.
Match the atomic numbers 4,8,10,15 and 19 with the following:
Element with four shells
Draw the atomic diagram of the following element showing the distribution of – protons, neutrons and the electrons in the various shell of the atom.
Carbon – \[\ce{^12_6C}\]
[The upper number represent the – mass number and the lower number represent the – atomic number e.g. calcium – mass number = 40, atomic number = 20]
‘Valency is the number of hydrogen atoms which can combine with [or displace] one atom of the element [or radical] forming a compound’. With reference to the above definition of valency, state the valency of chlorine in hydrogen chloride, giving reasons.
The diagram represents an isotope of hydrogen [H]. Answer the following:

Atomic no. = 1
Mass no. = 1
If an isotope of ‘H’ has mass no. = 3, how many neutrons does it have.
Which of the following are true for an element?
- Atomic number = number of protons + number of electrons
- Mass number = number of protons + number of neutrons
- Atomic mass = number of protons = number of neutrons
- Atomic number = number of protons = number of electrons
