Advertisements
Advertisements
प्रश्न
Draw two structural isomers of butane.
उत्तर
The two isomers of butane, C4H10 are:
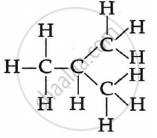
APPEARS IN
संबंधित प्रश्न
Select saturated hydrocarbons from the following: C3H6; C5H10; C4H10; C6H14; C2H4
Draw the structures for Ethanoic acid.
Draw the structures of possible isomers of butane, C4H10.
Give reason why the carbon compounds do not conduct electricity in the molten state.
Which of the two has a branched chain : isobutane or normal butane?
Give the name and structural formula of one member each of the following:
alkyne
An alkyne has seventy five carbon atoms in its molecule. The number of hydrogen atoms in its molecule will be:
(a) 150
(b) 148
(c) 152
(d) 146
The solid element A exhibits the property of catenation. It is also present in the form of a gas B in the air which is utilised by plants in photosynthesis. An allotrope C of this element is used in glass cutters.
(a) What is element A?
(b) What is the gas B?
(c) Name the allotrope C.
(d) State another use of allotrope C (other than in glass cutters).
(e) Name another allotrope of element A which exists as spherical molecules.
(f) Name a yet another allotrope of element A which conduct electricity.
Hydrocarbons are mainly composed of which of these?
How will you prove that C4H8 and C5H10 are homologues?
