Advertisements
Advertisements
Question
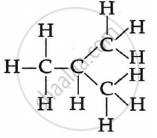
Draw two structural isomers of butane.
Solution
The two isomers of butane, C4H10 are:
\[\begin{array}{cc}
\ce{H}\phantom{...}\ce{H}\phantom{...}\ce{H}\phantom{...}\ce{H}\phantom{..}\\
|\phantom{....}|\phantom{....}|\phantom{....}|\phantom{..}\\
\ce{H - C - C - C - C - H}\\
|\phantom{....}|\phantom{....}|\phantom{....}|\phantom{..}\\
\ce{H}\phantom{...}\ce{H}\phantom{...}\ce{H}\phantom{...}\ce{H}\phantom{..}
\end{array}\]
APPEARS IN
RELATED QUESTIONS
Write the name and general formula of a chain of hydrocarbons in which an addition reaction with hydrogen is possible. State the essential condition for an addition reaction. Stating this condition, write a chemical equation giving the name of the reactant and the product of the reaction.
Fill in the following blank with suitable word:
CnH2n is the general formula of .......... hydrocarbons.
What is the unique property of carbon atom? How is this property helpful to us?
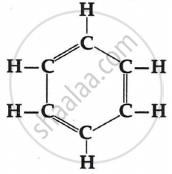
Giving their structures, state the number of single bonds, double bonds and triple bonds (if any) in the following compounds:
benzene
You are given the following molecular formulae of some hydrocarbons:
C5H8; C7H14; C6H6; C5H10; C7H12; C6H12
Which three formulae can represent cyclic hydrocarbons?
Write the molecular and structural formula of a cyclic hydrocarbon whose molecule contains 8 atoms of carbon.
A reagent which can help us to distinguish between alkenes and alkynes is ______.
Intake of small quantity of methanol can be lethal. Comment.
Recognise the carbon chain type of the following:
\[\begin{array}{cc}\ce{H}\phantom{...}\ce{H}\phantom{...}\ce{H}\\
|\phantom{....}|\phantom{....}|\\\ce{H - C - C - C - H}\\
|\phantom{....}|\phantom{....}|\\\ce{H}\phantom{...}\ce{}\phantom{.....}\ce{H}\\
\ce{H - C - H}\\
|\\
\ce{H}
\end{array}\]
Name the following: