Advertisements
Advertisements
प्रश्न
Draw two structural isomers of butane.
उत्तर
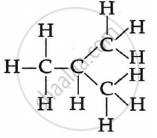
The two isomers of butane, C4H10 are:
\[\begin{array}{cc}
\ce{H}\phantom{...}\ce{H}\phantom{...}\ce{H}\phantom{...}\ce{H}\phantom{..}\\
|\phantom{....}|\phantom{....}|\phantom{....}|\phantom{..}\\
\ce{H - C - C - C - C - H}\\
|\phantom{....}|\phantom{....}|\phantom{....}|\phantom{..}\\
\ce{H}\phantom{...}\ce{H}\phantom{...}\ce{H}\phantom{...}\ce{H}\phantom{..}
\end{array}\]
APPEARS IN
संबंधित प्रश्न
List two reasons for carbon forming a large number of compounds. Name the type of bonding found in most of its compounds. Why does carbon form compounds mainly by this kind of bonding?
Draw the structures for Bromopentane*
Fill in the following blank with suitable word:
CnH2n is the general formula of .......... hydrocarbons.
Which of the two has a branched chain : isobutane or normal butane?
What is the unique property of carbon atom? How is this property helpful to us?
Two organic compounds A and B have the same molecular formula C6H12. Write the names and structural formulae:
Which compound contains single bonds as well as a double bond?
The functional group which always occurs in the middle of a carbon chain is:
(a) alcohol group
(b) aldehyde group
(c) carboxyl group
(d) ketone group
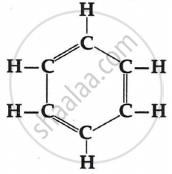
Recognise the carbon chain type of the following:
\[\begin{array}{cc}
\ce{H}\phantom{...}\ce{H}\phantom{...}\ce{H}\phantom{...}\ce{H}\\
|\phantom{....}|\phantom{....}|\phantom{....}|\\\ce{H - C - C - C - C - H}\\|\phantom{....}|\phantom{....}|\phantom{....}|\\
\ce{H}\phantom{...}\ce{H}\phantom{...}\ce{H}\phantom{...}\ce{H}
\end{array}\]
Name the following:
Draw the structure of propanone.
