Advertisements
Advertisements
प्रश्न
Explain the acidic nature of carboxylic acids.
उत्तर
Acidic nature of carboxylic acids:
- The carboxyl group (–COOH) imparts an acidic character to carboxylic acids.
- A carboxyl group is made of –OH group bonded to a carbonyl group.
- In aqueous solution, the H atom in OH of carboxyl group dissociates as proton and carboxylate ion is formed as the conjugate base,
\[\begin{array}{cc}
\phantom{.}\ce{O}\phantom{......................}\ce{O}\phantom{...........}\\
\phantom{.}||\phantom{......................}||\phantom{...........}\\
\ce{\underset{\text{Carboxylic acid}}{R - C -OH} + H2O ⇌ \underset{\text{Carboxylate ion}}{R - C - O-} + H3O+}
\end{array}\] - Carboxylate ion is resonance stabilized by two equivalent resonance structures as shown below.
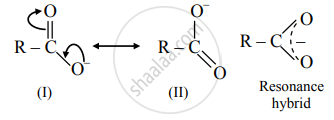
- Carboxylate ion has two resonance structures (I) and (II) and both of them are equivalent to each other.
- This gives good resonance stabilization to carboxylate ion, which in turn gives an acidic character to carboxylic acids.
APPEARS IN
संबंधित प्रश्न
Write the structure of the product formed when carboxylic acid is heated with a dehydrating agent like P2O5
Write the reducing agent which CANNOT reduce –COOH group.
What is the action of following on proponal?
Hydrazine
Write the preparation reactions for acid amide from the following.
Acid chloride
Write reactions for the following conversions.
Propanone to Propane
Write reactions for the following conversions.
4-Nitrobenzoic acid to Nitrobenzene
Write chemical reactions to convert –COOH group of acetic acid into the following.
CH4
Write chemical reactions to convert –COOH group of acetic acid into the following.
C2H5OH
Write reaction for preparation of acetophenone from benzoyl chloride.
Write reactions for the action of the following reagents on p-chlorobenzaldehyde.
Ethane-1,2-diol in presence of dry HCl.
Complete the following sequence of reactions and write structures for A, B, C.
\[\ce{Dry ice ->[i. Dry ether][ii. Hydrolysis] A ->[PCl5] B ->[H2(gas)][Pd - BaSO4] C}\]
In the reaction, \[\ce{C6H5COCH3 ->[{[H]}][Zn-Hg/conc. HCl] X}\], X is ______.
Which is the gas evolved when carboxylic acids react with strongly electropositive metals (such as Na, K, Ca, Zn)?
The product of the reaction between dialkyl cadmium and acyl chloride is a/an ____________.
Which of the following carboxylic acids will have the highest acidity?
The elimination of CO2 from a carboxylic acid is known as ____________.
Which among the following is the strongest acid?
The oxidation number of B in NaBH4 is ____________.
Which of the following functional groups is reduced by diborane?
Which of the following compounds is obtained when ethanoic anhydride is treated with water?
Which of the following carboxylic acids ha highest acidic strength?
Write structure of adipic acid.
Draw structures 01 conjugate bases of monochloroacetic acid and dichloroacetic acid. Which one is more stabilized by -I effect?
How will you prepare Acetic anhydride from acetic acid?
