Advertisements
Advertisements
प्रश्न
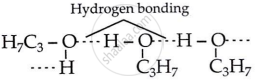
Explain why propanol has higher boiling point than that of the hydrocarbon, butane?
उत्तर
Propanol and butane have nearly the same molecular mass, but propanol has a higher boiling point because of intermolecular hydrogen bonding between its molecules. In butane, hydrogen bonding is not present due to the absence of the polar −OH group. The molecules of butane are connected by weaker van der Waals attraction forces.
APPEARS IN
संबंधित प्रश्न
Give reasons for the following: Butan-1-ol has a higher boiling point than diethyl ether.
Give reasons for the following : Phenol is more acidic than ethanol.
Alcohols are comparatively more soluble in water than hydrocarbons of comparable molecular masses. Explain this fact.
`CH_3-CH_2-Br"Alcoholic KOH"/""> B " HBR"/"">C"Na/Eather"/"">D`, the Compound D is
(A) ethane
(B) propane
(C) n-butane
(D) n-pentane
Write a chemical reaction to show that the open structure of D-glucose contains the following :
Five alcohol groups
Which one of the following alcohols is least soluble in water?
Arrange the following compounds in increasing order of boiling point. Propan-1-ol, butan-1-ol, butan-2-ol, pentan-1-ol
Explain why alcohols and ethers of comparable molecular mass have different boiling points?
Assertion: Boiling points of alcohols and ethers are high.
Reason: They can form intermolecular hydrogen-bonding.
Arrange the following in the increasing order of their property indicated:
ethanol, ethanoic acid, benzoic acid (boiling point)
Arrange the following in order of increasing boiling point:
Ethoxyethane, Butanal, Butanol, n-butane
Assertion (A): Alcohols react both as nucleophiles and electrophiles.
Reason (R): The bond between C–O is broken when alcohols react as nucleophiles.
Select the most appropriate answer from the options given below:
How are the following conversion carried out?
Methyl magnesium bromide →2-Methylpropan-2-ol.
What is esterifications? How is an ester obtained from alcohol or phenol?
How are the following conversions carried out?
Methyl magnesium bromide → 2 -Methylpropan-2-ol.
Convert the following:
acetaldehyde to isopropyl alcohol.
How are the following conversions carried out?
\[\ce{Methyl magnesium bromide ->2-Methylpropan-2-ol}\]
