Advertisements
Advertisements
प्रश्न
Fill in the following blank with suitable word:
CnH2n is the general formula of .......... hydrocarbons.
उत्तर
CnH2n is the general formula of alkene hydrocarbons.
APPEARS IN
संबंधित प्रश्न
What is meant by isomers?
Why does the element carbon from a large number of carbon compounds?
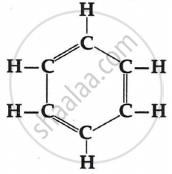
Write the molecular formula and structure of cyclohexane. How many covalent bonds are there in a molecule of cyclohexane?
An alkyne has seventy five carbon atoms in its molecule. The number of hydrogen atoms in its molecule will be:
(a) 150
(b) 148
(c) 152
(d) 146
Name the functional group which always occurs in the middle of a carbon chain.
The functional group which always occurs in the middle of a carbon chain is:
(a) alcohol group
(b) aldehyde group
(c) carboxyl group
(d) ketone group
Three organic compounds A, B and C have the following molecular formulae: C4H8O
Which molecular formula can represent an aldehyde as well as a ketone? Write the names and structural formulae of the aldehyde and ketone represented by this molecular formula.
Which of the following are correct structural isomers of butane?
- \[\begin{array}{cc}
\ce{H}\phantom{...}\ce{H}\phantom{...}\ce{H}\phantom{...}\ce{H}\phantom{..}\\
|\phantom{....}|\phantom{....}|\phantom{....}|\phantom{..}\\
\ce{H - C - C - C - C - H}\\
|\phantom{....}|\phantom{....}|\phantom{....}|\phantom{..}\\
\ce{H}\phantom{...}\ce{H}\phantom{...}\ce{H}\phantom{...}\ce{H}\phantom{..}
\end{array}\] - \[\begin{array}{cc}
\ce{H}\phantom{...}\ce{H}\phantom{...}\ce{H}\\
|\phantom{....}|\phantom{....}|\\
\ce{H - C - C - C - H}\\
|\phantom{.....}\backslash\phantom{..}|\\
\phantom{.....}\ce{H}\phantom{.......}\ce{C - H}\\
\phantom{.........}|\\
\phantom{.........}\ce{H}
\end{array}\] - \[\begin{array}{cc}
\ce{H}\phantom{...}\ce{H}\phantom{...}\ce{H}\\
|\phantom{....}|\phantom{....}|\\
\ce{H - C - C - C - H}\\
|\phantom{....}\phantom{....}|\\
\ce{H}\phantom{........}\ce{H}\\
|\\
\ce{H - C - H}\\
|\\\ce{H}
\end{array}\] - \[\begin{array}{cc}
\ce{H}\phantom{...}\ce{H}\\
|\phantom{....}|\\
\ce{H - C - C - H}\\
|\phantom{....}|\\
\ce{H - C - C - H}\\
|\phantom{....}|\\
\ce{H}\phantom{...}\ce{H}
\end{array}\]
Name the following:
Draw the structure of propanone.
