Advertisements
Advertisements
प्रश्न
Give appropriate scientific reasons for the following statement :
Carbon tetrachloride does not conduct electricity.
उत्तर
Carbon tetrachloride is a liquid and does not conduct electricity because it is a covalent compound and there are no free ions present and it contains only molecules.
APPEARS IN
संबंधित प्रश्न
Molten lead bromide conducts electricity. It is called an ______. It is composed of lead ______ and bromide ______. The lead ions are ______ charged and are called ______. The bromide ______ are ______ charged and are called ______.
Al+3 ,Cu+2 ,Na+ ,Zn+2 ions are present in aqueous solution, such that the concentration of ions is same, write the order of discharge of ions.
Choose the correct words to fill in the blanks.
Cations are formed by _______ (loss/ gain) of electrons and anions are formed by ________( loss/gain) of electrons.
Select the correct answer from the choices a,b,c and d which are given. Write only the letter corresponding to the correct answer.
During ionization metals lose electrons, this change can be called _______________.
Copper sulphate solution is electrolyzed using copper electrodes. Study the diagram given alongside and answer the questions that follow.
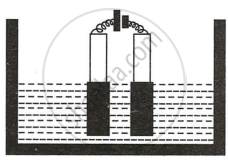
- Which electrode to your left or right is known as the oxidizing electrode and why?
- Write the equation representing the reaction that occurs.
- State two appropriate observations for the above electrolysis reactions.
Give appropriate scientific reasons for the following statement :
Zinc oxide can be reduced to zinc by using carbon monoxide, but aluminium oxide cannot be reduced by a reducing agent.
Name the metallic ions that should be present in the electrolyte when an article made copper is to be electroplated with silver
Select the ion, that would get selectively discharge from the aqueous mixture of the ions listed below :
\[\ce{SO^{2-}_{4}}\], \[\ce{NO^{-}_{3}}\], \[\ce{OH-}\]
Classify the following substance:
Ammonium hydroxide
Classify the following substance:
Dilute sulphuric acid
