Advertisements
Advertisements
प्रश्न
Give appropriate scientific reasons for the following statement :
Zinc oxide can be reduced to zinc by using carbon monoxide, but aluminium oxide cannot be reduced by a reducing agent.
उत्तर
Zinc is lower in the reactivity series, so it is comparatively less reactive. Hence, it is reduced by using carbon monoxide. But aluminium is very reactive; hence, it cannot be reduced by using a reducing agent and it can be reduced only by electrolytic reduction.
APPEARS IN
संबंधित प्रश्न
Identify the substance underlined, in the following case
The particles present in a liquid such as kerosene, that is a nonelectrolyte.
Complete the following by selecting the correct option from the choices given:
The divalent metal whose oxide is reduced to metal by electrolysis of its fused salt is __________. (Al/Na/Mg/K)
Give appropriate scientific reasons for During electrolysis of molten lead bromide, graphite anode is preferred to other electrodes.
State your observation At the cathode, when acidified aqueous copper sulphate solution is electrolysed with copper electrodes.
Define the term : Non-electrolyte
A solution of caustic soda (NaOH) in water or when fused, conducts an electric current. what is the similarity in these two cases?
If a fused metallic chloride is electrolyzed, at which electrode would the metal be obtained?
Copper sulphate solution is electrolyzed using copper electrodes. Study the diagram given alongside and answer the questions that follow.
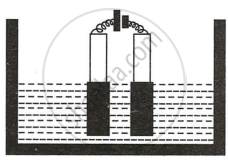
- Which electrode to your left or right is known as the oxidizing electrode and why?
- Write the equation representing the reaction that occurs.
- State two appropriate observations for the above electrolysis reactions.
A strip of copper is placed in four different colourless salt solutions. They are KNO3, AgNO3, Zn(NO3)2 and Ca(NO3)2. Which one of the solutions will finally turn blue?
Classify the following substance:
Ammonium hydroxide
