Advertisements
Advertisements
प्रश्न
Give reasons why Although copper is a good conductor of electricity, it is a non-electrolyte.
उत्तर
Copper has no mobile electrons in the solid state and an electrolyte should dissociate into oppositely charged ions to conduct electricity. Hence, copper is a non-electrolyte.
APPEARS IN
संबंधित प्रश्न
State the observations at the anode and at the cathode during the electrolysis of fused lead bromide using graphite electrodes
State the observations at the anode and at the cathode during the electrolysis of copper sulphate solution using copper electrodes.
A solution of HCl gas in water conducts electricity because ______, but a solution of HCl gas in toluene does not conduct an electric current because ______.
Amongst the OH- ions and Br- ions which are likely to discharge first?
A solution of caustic soda (NaOH) in water or when fused, conducts an electric current. what is the similarity in these two cases?
What is meant by the term 'electrolyte'?
If a fused metallic chloride is electrolyzed, at which electrode would the metal be obtained?
Why is carbon tetrachloride, which is a liquid, a non-electrolyte?
Copper sulphate solution is electrolyzed using copper electrodes. Study the diagram given alongside and answer the questions that follow.
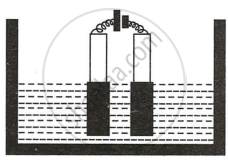
- Which electrode to your left or right is known as the oxidizing electrode and why?
- Write the equation representing the reaction that occurs.
- State two appropriate observations for the above electrolysis reactions.
Classify the following substance:
Dilute hydrochloric acid
