Advertisements
Advertisements
प्रश्न
Give the main experimental points only to demonstrate that magnesium.
उत्तर
Experiment to show that magnesium gains weight on burning:
Take dry crucible with lid. Put a small piece of magnesium in it and cover the lid. Find its weight with lid covering it. Let it be 50 gms.
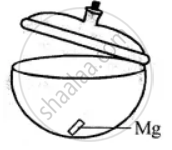
Now heat the crucible along with magnesium and lid just opened for a while. Taking care that no ash comes out. Let it cool covering the lid and products formed weight it again. The weight now will be more. Let it be 50.82 gms, oxygen combined is 50.82 – 50 = 0.82 gms this shows that something is added to magnesium, which has increased its weight. This 0.82 gm is the weight of oxygen added to magnesium.
2Mg + O2 → 2MgO
Hence magnesium gains weight on burning.
APPEARS IN
संबंधित प्रश्न
A mixture of oxygen and carbon dioxide used for artificial respiration.
What is rust ?
Write ‘True’ or ‘False’ in front of following statement.
Nitrogen is a gaseous non-metal essential for respiration.
What happens when Sodium burns in oxygen ? Also give the balanced chemical equation for the reaction.
Write the balanced chemical equation of the manganese dioxide.
State how nitrogen is utilised by plants for their growth. Give a reason why nitrogen is used in flushing food packages.
Describe simple experiments to show the presence of -
oxygen and nitrogen component in air using a bell jar
Give reason for the following:
Acid rain has caused damage to heritage structures such as the Taj Mahal.
Name the following:
The pollutant which combines with water vapour to give sulphuric acid – a product of acid rain.
What is meant by burning?
