Advertisements
Advertisements
प्रश्न
Given the standard electrode potentials,
\[\ce{K+/K}\] = −2.93 V, \[\ce{Ag+/Ag}\] = 0.80 V,
\[\ce{Hg^{2+}/Hg}\] = 0.79 V
\[\ce{Mg^{2+}/Mg}\] = −2.37 V, \[\ce{Cr^{3+}/Cr}\] = −0.74 V
Arrange these metals in their increasing order of reducing power.
उत्तर
The reducing power of a metal depends on its oxidation potential. The higher the oxidation potential, the greater its tendency to be oxidized and, hence, the greater its reducing power. Hence the order of increasing of reducing the power of the given metals will be as follows:
\[\ce{Ag < Hg < Cr < Mg < K}\]
APPEARS IN
संबंधित प्रश्न
Arrange the following reducing agents in the order of increasing strength under standard state conditions. Justify the answer
|
Element |
Al(s) |
Cu(s) |
Cl(aq) |
Ni(s) |
|
Eo |
-1.66V |
0.34V |
1.36V |
-0.26V |
Can copper sulphate solution be stored in an iron vessel? Explain.
The standard e.m.f of the following cell is 0.463 V
`Cu|Cu_(1m)^(++)`
What is the standard potential of Cu electrode?
(A) 1.137 V
(B) 0.337 V
(C) 0.463 V
(D) - 0.463 V
Calculate emf of the following cell at 25°C:
\[\ce{Sn/Sn^2+ (0.001 M) || H+ (0.01 M) | H2_{(g)} (1 bar) | Pt_{(s)}}\]
Given: \[\ce{E^\circ(Sn^2+/sn) = -0.14 V, E^\circ H+/H2 = 0.00 V (log 10 = 1)}\]
Calculate e.m.f of the following cell at 298 K:
2Cr(s) + 3Fe2+ (0.1M) → 2Cr3+ (0.01M) + 3 Fe(s)
Given: E°(Cr3+ | Cr) = – 0.74 VE° (Fe2+ | Fe) = – 0.44 V
Calculate the emf of the following cell at 25°C :
Standard electrode potential is measured taking the concentrations of all the species involved in a half-cell is ____________.
The difference between the electrode potentials of two electrodes when no current is drawn through the cell is called ______.
The positive value of the standard electrode potential of Cu2+/Cu indicates that:
(i) this redox couple is a stronger reducing agent than the H+/H2 couple.
(ii) this redox couple is a stronger oxidising agent than H+/H2 .
(iii) Cu can displace H2 from acid.
(iv) Cu cannot displace H2 from acid.
What does the negative sign in the expression `"E"^Θ ("Zn"^(2+))//("Zn")` = − 0.76 V mean?
Value of standard electrode potential for the oxidation of \[\ce{Cl-}\] ions is more positive than that of water, even then in the electrolysis of aqueous sodium chloride, why is \[\ce{Cl-}\] oxidised at anode instead of water?
Which reference electrode is used to measure the electrode potential of other electrodes?
Consider the figure and answer the following question.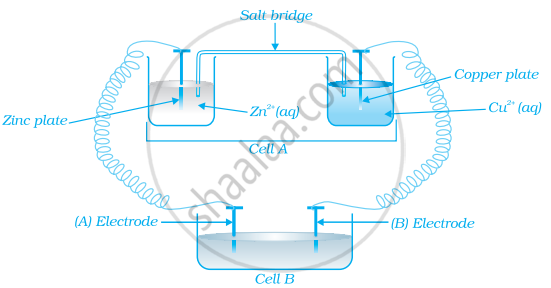
If cell ‘A’ has ECell = 0.5V and cell ‘B’ has ECell = 1.1V then what will be the reactions at anode and cathode?
Represent the cell in which the following reaction takes place.The value of E˚ for the cell is 1.260 V. What is the value of Ecell?
\[\ce{2Al (s) + 3Cd^{2+} (0.1M) -> 3Cd (s) + 2Al^{3+} (0.01M)}\]
Standard electrode potential of three metals X, Y and Z are – 1.2 V, + 0.5 V and – 3.0 V, respectively. The reducing power of these metals will be:
The potential of a hydrogen electrode at PH = 10 is
Standard reduction potentials (E°) of Cd2+, respectively which is the strongest reducing agent
